Kynurenine aminotransferase 3/glutamine transaminase L/cysteine conjugate beta-lyase 2 is a major glutamine transaminase in the mouse kidney
- PMID: 28955961
- PMCID: PMC5613967
- DOI: 10.1016/j.bbrep.2016.09.008
Kynurenine aminotransferase 3/glutamine transaminase L/cysteine conjugate beta-lyase 2 is a major glutamine transaminase in the mouse kidney
Abstract
Background: Kynurenine aminotransferase 3 (KAT3) catalyzes the transamination of Kynurenine to kynurenic acid, and is identical to cysteine conjugate beta-lyase 2 (CCBL2) and glutamine transaminase L (GTL). GTL was previously purified from the rat liver and considered as a liver type glutamine transaminase. However, because of the substrate overlap and high sequence similarity of KAT3 and KAT1, it was difficult to assay the specific activity of each KAT and to study the enzyme localization in animals.
Methods: KAT3 transcript and protein levels as well as enzyme activity in the liver and kidney were analyzed by regular reverse transcription-polymerase chain reaction (RT-PCR), real time RT-PCR, biochemical activity assays combined with a specific inhibition assay, and western blotting using a purified and a highly specific antibody, respectively.
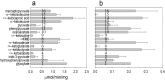
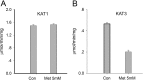
Results: This study concerns the comparative biochemical characterization and localization of KAT 3 in the mouse. The results showed that KAT3 was present in both liver and kidney of the mouse, but was much more abundant in the kidney than in the liver. The mouse KAT3 is more efficient in transamination of glutamine with indo-3-pyruvate or oxaloacetate as amino group acceptor than the mouse KAT1.
Conclusions: Mouse KAT3 is a major glutamine transaminase in the kidney although it was named a liver type transaminase.
General significance: Our data highlights KAT3 as a key enzyme for studying the nephrotoxic mechanism of some xenobiotics and the formation of chemopreventive compounds in the mouse kidney. This suggests tissue localizations of KAT3/GTL/CCBL2 in other animals may be carefully checked.
Keywords: Aminotransferase; CCBL, cysteine conjugate beta-lyase; Cysteine conjugate beta-lyase 2; GTK, glutamine transaminase K; GTL, glutamine transaminase L; Glutamine transaminase; KAT, kynurenine aminotransferase; KYNA, kynurenic acid; Kynurenic acid; Kynurenine; Kynurenine aminotransferase; PLP, pyridoxal-5′-phosphate..
Figures



Similar articles
-
Kynurenine aminotransferase III and glutamine transaminase L are identical enzymes that have cysteine S-conjugate β-lyase activity and can transaminate L-selenomethionine.J Biol Chem. 2014 Nov 7;289(45):30950-61. doi: 10.1074/jbc.M114.591461. Epub 2014 Sep 17. J Biol Chem. 2014. PMID: 25231977 Free PMC article.
-
The role of glutamine transaminase K (GTK) in sulfur and alpha-keto acid metabolism in the brain, and in the possible bioactivation of neurotoxicants.Neurochem Int. 2004 Jun;44(8):557-77. doi: 10.1016/j.neuint.2003.12.002. Neurochem Int. 2004. PMID: 15016471 Review.
-
Cloning and expression of a rat kidney cytosolic glutamine transaminase K that has strong sequence homology to kynurenine pyruvate aminotransferase.Arch Biochem Biophys. 1996 Nov 15;335(2):311-20. doi: 10.1006/abbi.1996.0512. Arch Biochem Biophys. 1996. PMID: 8914928
-
Glutamine transaminase K and cysteine S-conjugate beta-lyase activity stains.Anal Biochem. 1991 Sep 2;197(2):421-7. doi: 10.1016/0003-2697(91)90414-o. Anal Biochem. 1991. PMID: 1723851
-
Structure, expression, and function of kynurenine aminotransferases in human and rodent brains.Cell Mol Life Sci. 2010 Feb;67(3):353-68. doi: 10.1007/s00018-009-0166-4. Epub 2009 Oct 15. Cell Mol Life Sci. 2010. PMID: 19826765 Free PMC article. Review.
Cited by
-
Kynurenic Acid Acts as a Signaling Molecule Regulating Energy Expenditure and Is Closely Associated With Metabolic Diseases.Front Endocrinol (Lausanne). 2022 Feb 24;13:847611. doi: 10.3389/fendo.2022.847611. eCollection 2022. Front Endocrinol (Lausanne). 2022. PMID: 35282457 Free PMC article. Review.
-
Upregulation of the Glutaminase II Pathway Contributes to Glutamate Production upon Glutaminase 1 Inhibition in Pancreatic Cancer.Proteomics. 2019 Nov;19(21-22):e1800451. doi: 10.1002/pmic.201800451. Epub 2019 Aug 1. Proteomics. 2019. PMID: 31231915 Free PMC article.
-
Pyridoxal 5'-Phosphate-Dependent Enzymes at the Crossroads of Host-Microbe Tryptophan Metabolism.Int J Mol Sci. 2020 Aug 13;21(16):5823. doi: 10.3390/ijms21165823. Int J Mol Sci. 2020. PMID: 32823705 Free PMC article. Review.
-
Cysteine conjugate beta-lyase 2 (CCBL2) expression as a prognostic marker of survival in breast cancer patients.PLoS One. 2022 Jun 30;17(6):e0269998. doi: 10.1371/journal.pone.0269998. eCollection 2022. PLoS One. 2022. PMID: 35771747 Free PMC article.
-
The Biology and Biochemistry of Kynurenic Acid, a Potential Nutraceutical with Multiple Biological Effects.Int J Mol Sci. 2024 Aug 21;25(16):9082. doi: 10.3390/ijms25169082. Int J Mol Sci. 2024. PMID: 39201768 Free PMC article. Review.
References
-
- Leeson P.D., Iversen L.L. The glycine site on the NMDA receptor: structure-activity relationships and therapeutic potential. J. Med. Chem. 1994;37:4053–4067. - PubMed
-
- Perkins M.N., Stone T.W. An iontophoretic investigation of the actions of convulsant kynurenines and their interaction with the endogenous excitant quinolinic acid. Brain Res. 1982;247:184–187. - PubMed
-
- Stone T.W., Perkins M.N. Actions of excitatory amino acids and kynurenic acid in the primate hippocampus: a preliminary study. Neurosci. Lett. 1984;52:335–340. - PubMed
-
- Birch P.J., Grossman C.J., Hayes A.G. Kynurenic acid antagonises responses to NMDA via an action at the strychnine-insensitive glycine receptor. Eur. J. Pharmcol. 1988;154:85–87. - PubMed
-
- Pereira E.F., Hilmas C., Santos M.D., Alkondon M., Maelicke A., Albuquerque E.X. Unconventional ligands and modulators of nicotinic receptors. J. Neurobiol. 2002;53:479–500. - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

