Blood brain barrier permeability of (-)-epigallocatechin gallate, its proliferation-enhancing activity of human neuroblastoma SH-SY5Y cells, and its preventive effect on age-related cognitive dysfunction in mice
- PMID: 28956003
- PMCID: PMC5614586
- DOI: 10.1016/j.bbrep.2016.12.012
Blood brain barrier permeability of (-)-epigallocatechin gallate, its proliferation-enhancing activity of human neuroblastoma SH-SY5Y cells, and its preventive effect on age-related cognitive dysfunction in mice
Abstract
Background: The consumption of green tea catechins (GTCs) suppresses age-related cognitive dysfunction in mice. GTCs are composed of several catechins, of which epigallocatechin gallate (EGCG) is the most abundant, followed by epigallocatechin (EGC). Orally ingested EGCG is hydrolyzed by intestinal biota to EGC and gallic acid (GA). To understand the mechanism of action of GTCs on the brain, their permeability of the blood brain barrier (BBB) as well as their effects on cognitive function in mice and on nerve cell proliferation in vitro were examined.
Methods: The BBB permeability of EGCG, EGC and GA was examined using a BBB model kit. SAMP10, a mouse model of brain senescence, was used to test cognitive function in vivo. Human neuroblastoma SH-SY5Y cells were used to test nerve cell proliferation and differentiation.
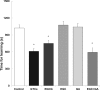
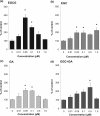
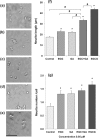
Results: The in vitro BBB permeability (%, in 30 min) of EGCG, EGC and GA was 2.8±0.1, 3.4±0.3 and 6.5±0.6, respectively. The permeability of EGCG into the BBB indicates that EGCG reached the brain parenchyma even at a very low concentration. The learning ability of SAMP10 mice that ingested EGCG (20 mg/kg) was significantly higher than of mice that ingested EGC or GA. However, combined ingestion of EGC and GA showed a significant improvement comparable to EGCG. SH-SY5Y cell growth was significantly enhanced by 0.05 µM EGCG, but this effect was reduced at higher concentrations. The effect of EGC and GA was lower than that of EGCG at 0.05 µM. Co-administration of EGC and GA increased neurite length more than EGC or GA alone.
Conclusion: Cognitive dysfunction in mice is suppressed after ingesting GTCs when a low concentration of EGCG is incorporated into the brain parenchyma via the BBB. Nerve cell proliferation/differentiation was enhanced by a low concentration of EGCG. Furthermore, the additive effect of EGC and GA suggests that EGCG sustains a preventive effect after the hydrolysis to EGC and GA.
Keywords: (−)-epigallocatechin gallate; 8-oxodG, 8-oxodeoxyguanosine; BBB, blood-brain barrier; Blood-brain barrier permeability; Brain plasticity; C, (+)-catechin; Cognitive dysfunction; EC, (−)-epicatechin; EGC, (−)-epigallocatechin; EGCG, (−)-epigallocatechin gallate; GA, gallic acid; GTC, green tea catechin; Green tea catechin; LC-MS/MS, liquid chromatography tandem-mass spectrometry; LPO, lipid peroxidation; MRM, multiple reaction-monitoring; Nerve cell proliferation; SAMP10, senescence-accelerated mouse prone 10..
Figures


References
-
- Unno K., Takabayashi F., Kishido T., Oku N. Suppressive effect of green tea catechins on morphologic and functional regression of the brain in aged mice with accelerated senescence (SAMP10) Exp Gerontol. 2004;39:1027–1034. - PubMed
-
- Unno K., Takabayashi F., Yoshida H., Choba D., Fukutomi R., Kikunaga N., Kishido T., Oku N., Hoshino M. Daily consumption of green tea catechin delays memory regression in aged mice. Biogerontology. 2007;8:89–95. - PubMed
-
- Unno K., Ishikawa Y., Takabayashi F., Sasaki T., Takamori N., Iguchi K., Hoshino M. Daily ingestion of green tea catechins from adulthood suppressed brain dysfunction in aged mice. Biofactors. 2008;34:263–271. - PubMed
-
- Shimada A., Keino H., Satoh M., Kishikawa M., Seriu N., Hosokawa M. Age-related progressive neuronal DNA damage associated with cerebral degeneration in a mouse model of accelerated senescence. J. Gerontol. A Biol. Sci. Med. Sci. 2002;57:B415–B421. - PubMed
-
- Shimada A., Keino H., Satoh M., Kishikawa M., Hosokawa M. Age-related loss of synapses in the frontal cortex of SAMP10 mouse: a model of cerebral degeneration. Synapse. 2003;48:198–204. - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources

