Specific titin and myomesin domains stimulate myoblast proliferation
- PMID: 28956009
- PMCID: PMC5614584
- DOI: 10.1016/j.bbrep.2016.12.007
Specific titin and myomesin domains stimulate myoblast proliferation
Abstract
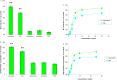
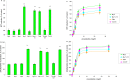
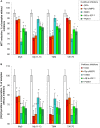
Myofibrillar proteins titin and myomesin stimulated myoblast proliferation as determined by MTT-test and labelled thymidine incorporation in the DNA. Specific Fn type III and Ig-like domains of these proteins were able to exert mitogenic effects as well. Proliferative effect of Fn type III domains was highly sensitive to inhibition of Ca2+/calmodulin dependent protein kinase, whereas the effect of Ig-like domains showed greater sensitivity to the inhibition of adenylyl cyclase - cAMP - PKA pathway. IGF-1 autocrine signalling inhibition partially suppressed mitogenic effects revealed by both domain types.
Keywords: Fn type III domain; IGF-1; Ig-like domain; Myoblast proliferation; Myomesin; Titin.
Figures
References
-
- Schiaffino S., Dyar K.A., Ciciliot S., Blaauw B., Sandri M. Mechanisms regulating skeletal muscle growth and atrophy. FEBS J. 2013;280:4294–4314. - PubMed
-
- Clemmons D.R. Role of IGF-I in skeletal muscle mass maintenance. Trends Endocrinol. Metab. 2009;20:349–356. - PubMed
-
- Banerjee A., Guttridge D.C. Mechanisms for maintaining muscle. Curr. Opin. Support Palliat. Care. 2012;6:451–456. - PubMed
-
- Yang S.Y., Goldspink G. Different roles of the IGF-I Ec peptide (MGF) and mature IGF-I in myoblast proliferation and differentiation. FEBS Lett. 2002;522:156–160. - PubMed
-
- Kandalla P.K., Goldspink G., Butler-Browne G., Mouly V. Mechano Growth Factor E peptide (MGF-E), derived from an isoform of IGF-1, activates human muscle progenitor cells and induces an increase in their fusion potential at different ages. Mech. Ageing Dev. 2011;132:154–162. - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

