Endothelium-specific amyloid precursor protein deficiency causes endothelial dysfunction in cerebral arteries
- PMID: 28959912
- PMCID: PMC6168907
- DOI: 10.1177/0271678X17735418
Endothelium-specific amyloid precursor protein deficiency causes endothelial dysfunction in cerebral arteries
Abstract
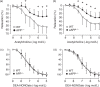
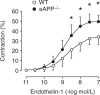
The exact physiological function of amyloid-β precursor protein (APP) in endothelial cells is unknown. Endothelium-specific APP-deficient (eAPP-/-) mice were created to gain new insights into the role of APP in the control of vascular endothelial function. Endothelium-dependent relaxations to acetylcholine were significantly impaired in basilar arteries of global APP knockout (APP-/-) and eAPP-/- mice ( P < 0.05). In contrast, endothelium-independent relaxations to nitric oxide (NO)-donor diethylamine-NONOate were unchanged. Western blot analysis revealed that protein expression of endothelial nitric oxide synthase (eNOS) was significantly downregulated in large cerebral arteries of APP-/- mice and eAPP-/- mice as compared to respective wild-type littermates ( P < 0.05). Furthermore, basal levels of cyclic guanosine monophosphate (cGMP) were also significantly reduced in large cerebral arteries of APP-deficient mice ( P < 0.05). In contrast, protein expression of prostacyclin synthase as well as levels of cyclic adenosine monophosphate (cAMP) was not affected by genetic inactivation of APP in endothelial cells. By using siRNA to knockdown APP in cultured human brain microvascular endothelial cells we also found a significant downregulation of eNOS mRNA and protein expressions in APP-deficient endothelium ( P < 0.05). These findings indicate that under physiological conditions, expression of APP in cerebral vascular endothelium plays an important protective function by maintaining constitutive expression of eNOS .
Keywords: Amyloid precursor protein; amyloid-β; cerebral arteries; endothelial nitric oxide synthase; endothelium.
Figures




Similar articles
-
Inactivation of BACE1 increases expression of endothelial nitric oxide synthase in cerebrovascular endothelium.J Cereb Blood Flow Metab. 2022 Oct;42(10):1920-1932. doi: 10.1177/0271678X221105683. Epub 2022 Jun 8. J Cereb Blood Flow Metab. 2022. PMID: 35673977 Free PMC article.
-
BACE2 deficiency impairs expression and function of endothelial nitric oxide synthase in brain endothelial cells.J Neurochem. 2023 Sep;166(6):928-942. doi: 10.1111/jnc.15929. Epub 2023 Aug 7. J Neurochem. 2023. PMID: 37547981 Free PMC article.
-
Vascular phenotype of amyloid precursor protein-deficient mice.Am J Physiol Heart Circ Physiol. 2019 Jun 1;316(6):H1297-H1308. doi: 10.1152/ajpheart.00539.2018. Epub 2019 Mar 22. Am J Physiol Heart Circ Physiol. 2019. PMID: 30901278 Free PMC article.
-
Endothelial nitric oxide: protector of a healthy mind.Eur Heart J. 2014 Apr;35(14):888-94. doi: 10.1093/eurheartj/eht544. Epub 2013 Dec 18. Eur Heart J. 2014. PMID: 24357508 Free PMC article. Review.
-
Expression and Processing of Amyloid Precursor Protein in Vascular Endothelium.Physiology (Bethesda). 2017 Jan;32(1):20-32. doi: 10.1152/physiol.00021.2016. Physiology (Bethesda). 2017. PMID: 27927802 Free PMC article. Review.
Cited by
-
Mouse models of Alzheimer's disease cause rarefaction of pial collaterals and increased severity of ischemic stroke.Angiogenesis. 2019 May;22(2):263-279. doi: 10.1007/s10456-018-9655-0. Epub 2018 Dec 5. Angiogenesis. 2019. PMID: 30519973 Free PMC article.
-
New Insights Into Blood-Brain Barrier Maintenance: The Homeostatic Role of β-Amyloid Precursor Protein in Cerebral Vasculature.Front Physiol. 2020 Aug 27;11:1056. doi: 10.3389/fphys.2020.01056. eCollection 2020. Front Physiol. 2020. PMID: 32973564 Free PMC article. Review.
-
Emerging Roles of Endothelial Nitric Oxide in Preservation of Cognitive Health.Stroke. 2023 Mar;54(3):686-696. doi: 10.1161/STROKEAHA.122.041444. Epub 2023 Feb 27. Stroke. 2023. PMID: 36848426 Free PMC article. Review.
-
Lung endothelium, tau, and amyloids in health and disease.Physiol Rev. 2024 Apr 1;104(2):533-587. doi: 10.1152/physrev.00006.2023. Epub 2023 Aug 10. Physiol Rev. 2024. PMID: 37561137 Free PMC article. Review.
-
Association of cerebral microvascular dysfunction and white matter injury in Alzheimer's disease.Geroscience. 2022 Aug;44(4):1-14. doi: 10.1007/s11357-022-00585-5. Epub 2022 May 25. Geroscience. 2022. PMID: 35612774 Free PMC article.
References
-
- Faraci FM, Heistad DD. Regulation of large cerebral arteries and cerebral microvascular pressure. Circ Res 1990; 66: 8–17. - PubMed
-
- Moncada S, Palmer RM, Higgs EA. Nitric oxide: physiology, pathophysiology, and pharmacology. Pharmacol Rev 1991; 43: 109–142. - PubMed
-
- Atochin DN, Demchenko IT, Astern J, et al. Contributions of endothelial and neuronal nitric oxide synthases to cerebrovascular responses to hyperoxia. J Cereb Blood Flow Metab 2003; 23: 1219–1226. - PubMed
-
- Ma J, Meng W, Ayata C, et al. L-NNA-sensitive regional cerebral blood flow augmentation during hypercapnia in type III NOS mutant mice. Am J Physiol 1996; 271: H1717–1719. - PubMed
-
- Sobey CG, Faraci FM. Effects of a novel inhibitor of guanylyl cyclase on dilator responses of mouse cerebral arterioles. Stroke 1997; 28: 837–842; discussion 842–833. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

