Cancer Immunotherapy Using CAR-T Cells: From the Research Bench to the Assembly Line
- PMID: 28960810
- PMCID: PMC5966018
- DOI: 10.1002/biot.201700097
Cancer Immunotherapy Using CAR-T Cells: From the Research Bench to the Assembly Line
Abstract
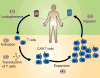
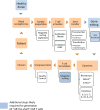
The focus of cancer treatment has recently shifted toward targeted therapies, including immunotherapy, which allow better individualization of care and are hoped to increase the probability of success for patients. Specifically, T cells genetically modified to express chimeric antigen receptors (CARs; CAR-T cells) have generated exciting results. Recent clinical successes with this cutting-edge therapy have helped to push CAR-T cells toward approval for wider use. However, several limitations need to be addressed before the widespread use of CAR-T cells as a standard treatment. Here, a succinct background on adoptive T-cell therapy (ATCT)is given. A brief overview of the structure of CARs, how they are introduced into T cells, and how CAR-T cell expansion and selection is achieved in vitro is then presented. Some of the challenges in CAR design are discussed, as well as the difficulties that arise in large-scale CAR-T cell manufacture that will need to be addressed to achieve successful commercialization of this type of cell therapy. Finally, developments already on the horizon are discussed.
Keywords: T cells; chimeric antigen receptors; genetic engineering; immunotherapy; manufacturing.
© 2017 WILEY-VCH Verlag GmbH & Co. KGaA, Weinheim.
Conflict of interest statement
The authors declare no financial or commercial conflict of interest.
Figures

References
-
- Seliger B. Journal of Immunotoxicology. 2008;5:361. - PubMed
-
- Ritchie DS, Neeson PJ, Khot A, Peinert S, Tai T, Tainton K, Chen K, Shin M, Wall DM, Hönemann D, Gambell P, Westerman DA, Haurat J, Westwood JA, Scott AM, Kravets L, Dickinson M, Trapani JA, Smyth MJ, Darcy PK, Kershaw MH, Prince HM. Molecular therapy : the journal of the American Society of Gene Therapy. 2013;21:2122. - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

