Long-term safety of human retinal progenitor cell transplantation in retinitis pigmentosa patients
- PMID: 28962643
- PMCID: PMC5622579
- DOI: 10.1186/s13287-017-0661-8
Long-term safety of human retinal progenitor cell transplantation in retinitis pigmentosa patients
Abstract
Background: Retinitis pigmentosa is a common genetic disease that causes retinal degeneration and blindness for which there is currently no curable treatment available. Vision preservation was observed in retinitis pigmentosa animal models after retinal stem cell transplantation. However, long-term safety studies and visual assessment have not been thoroughly tested in retinitis pigmentosa patients.
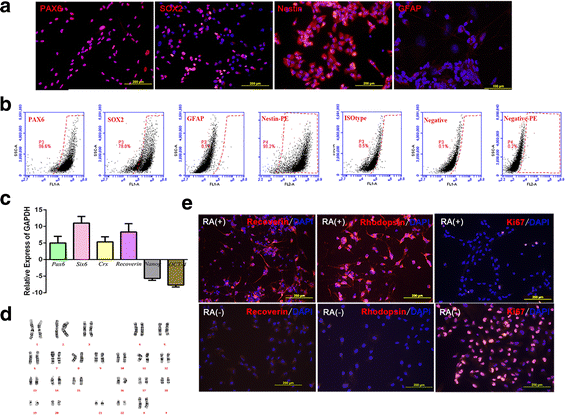
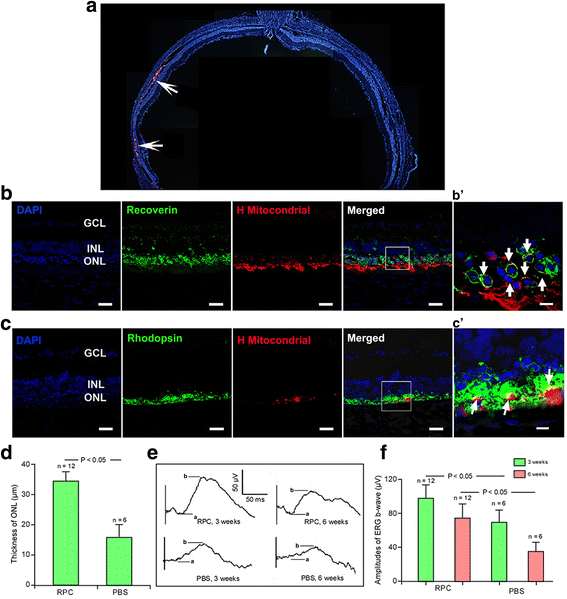
Methods: In our pre-clinical study, purified human fetal-derived retinal progenitor cells (RPCs) were transplanted into the diseased retina of Royal College of Surgeons (RCS) rats, a model of retinal degeneration. Based on these results, we conducted a phase I clinical trial to establish the safety and tolerability of transplantation of RPCs in eight patients with advanced retinitis pigmentosa. Patients were studied for 24 months.
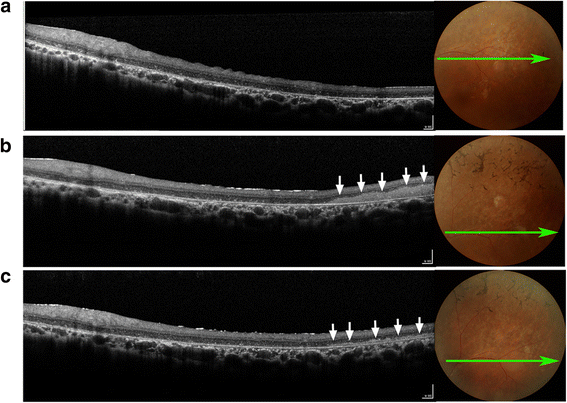
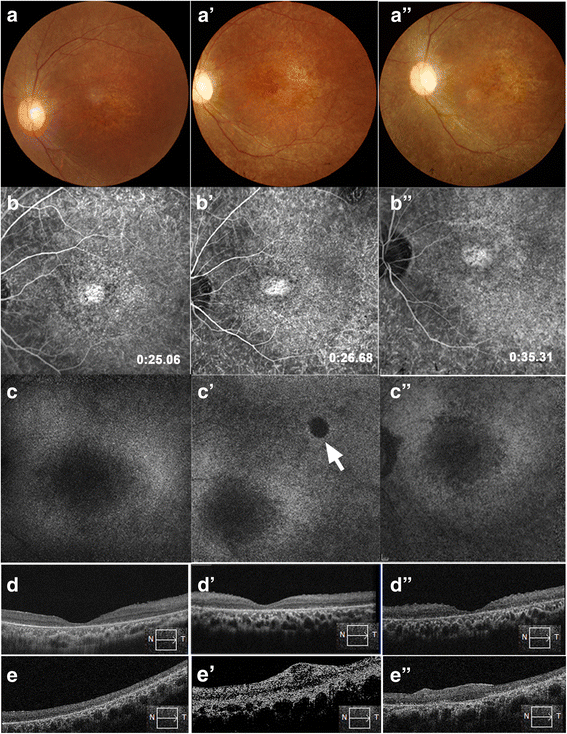
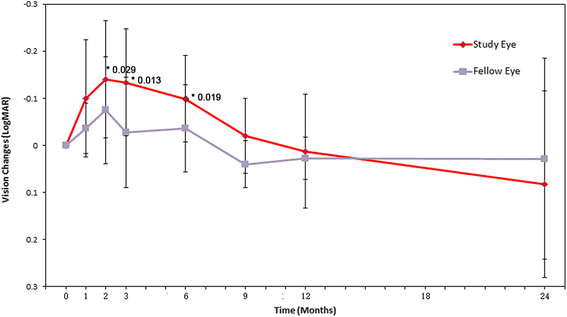
Results: After RPC transplantation in RCS rats, we observed moderate recovery of vision and maintenance of the outer nuclear layer thickness. Most importantly, we did not find tumor formation or immune rejection. In the retinis pigmentosa patients given RPC injections, we also did not observe immunological rejection or tumorigenesis when immunosuppressive agents were not administered. We observed a significant improvement in visual acuity (P < 0.05) in five patients and an increase in retinal sensitivity of pupillary responses in three of the eight patients between 2 and 6 months after the transplant, but this improvement did not appear by 12 months.
Conclusion: Our study for the first time confirmed the long-term safety and feasibility of vision repair by stem cell therapy in patients blinded by retinitis pigmentosa.
Trial registration: WHO Trial Registration, ChiCTR-TNRC-08000193 . Retrospectively registered on 5 December 2008.
Keywords: Cell transplantation; Progenitor cell; Retina; Retinitis pigmentosa; Visual improvement.
Conflict of interest statement
Ethics approval and consent to participate
The trial was conducted at the Southwest Hospital, Chongqing, China, and was approved by the Medical Ethics Committee of Southwest Hospital, Third Military Medical University. The research adhered to the principles of the Declaration of Helsinki, and all participants provided their written informed consent and surgical consent and approved the procedure for publishing our studies (WHO Trial Registration, ChiCTR-TNRC-08000193).
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Figures
Similar articles
-
Combined transplantation of human mesenchymal stem cells and human retinal progenitor cells into the subretinal space of RCS rats.Sci Rep. 2017 Mar 15;7(1):199. doi: 10.1038/s41598-017-00241-5. Sci Rep. 2017. PMID: 28298640 Free PMC article.
-
Human primitive mesenchymal stem cell-derived retinal progenitor cells improved neuroprotection, neurogenesis, and vision in rd12 mouse model of retinitis pigmentosa.Stem Cell Res Ther. 2022 Apr 8;13(1):148. doi: 10.1186/s13287-022-02828-w. Stem Cell Res Ther. 2022. PMID: 35395806 Free PMC article.
-
Transplantation of rat embryonic stem cell-derived retinal cells restores visual function in the Royal College of Surgeons rats.Doc Ophthalmol. 2018 Oct;137(2):71-78. doi: 10.1007/s10633-018-9648-8. Epub 2018 Aug 3. Doc Ophthalmol. 2018. PMID: 30074097
-
Stem cell transplantation as a progressing treatment for retinitis pigmentosa.Cell Tissue Res. 2022 Feb;387(2):177-205. doi: 10.1007/s00441-021-03551-3. Epub 2022 Jan 10. Cell Tissue Res. 2022. PMID: 35001210 Review.
-
Cell replacement and visual restoration by retinal sheet transplants.Prog Retin Eye Res. 2012 Nov;31(6):661-87. doi: 10.1016/j.preteyeres.2012.06.003. Epub 2012 Jul 5. Prog Retin Eye Res. 2012. PMID: 22771454 Free PMC article. Review.
Cited by
-
Subretinal Injection Techniques for Retinal Disease: A Review.J Clin Med. 2022 Aug 12;11(16):4717. doi: 10.3390/jcm11164717. J Clin Med. 2022. PMID: 36012955 Free PMC article. Review.
-
Novel stem cell and gene therapy in diabetic retinopathy, age related macular degeneration, and retinitis pigmentosa.Int J Retina Vitreous. 2019 Feb 13;5:7. doi: 10.1186/s40942-019-0158-y. eCollection 2019. Int J Retina Vitreous. 2019. PMID: 30805203 Free PMC article. Review.
-
Cell Ferroptosis: New Mechanism and New Hope for Retinitis Pigmentosa.Cells. 2021 Aug 21;10(8):2153. doi: 10.3390/cells10082153. Cells. 2021. PMID: 34440922 Free PMC article. Review.
-
The Future of Regenerative Medicine: Cell Therapy Using Pluripotent Stem Cells and Acellular Therapies Based on Extracellular Vesicles.Cells. 2021 Jan 27;10(2):240. doi: 10.3390/cells10020240. Cells. 2021. PMID: 33513719 Free PMC article. Review.
-
Next-generation stem cells - ushering in a new era of cell-based therapies.Nat Rev Drug Discov. 2020 Jul;19(7):463-479. doi: 10.1038/s41573-020-0064-x. Epub 2020 Apr 6. Nat Rev Drug Discov. 2020. PMID: 32612263 Review.
References
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources

