Yellow Fever Virus, but Not Zika Virus or Dengue Virus, Inhibits T-Cell Receptor-Mediated T-Cell Function by an RNA-Based Mechanism
- PMID: 28968905
- PMCID: PMC5853456
- DOI: 10.1093/infdis/jix462
Yellow Fever Virus, but Not Zika Virus or Dengue Virus, Inhibits T-Cell Receptor-Mediated T-Cell Function by an RNA-Based Mechanism
Abstract
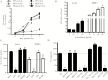
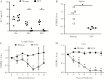
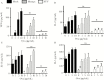
The Flavivirus genus within the Flaviviridae family is comprised of many important human pathogens including yellow fever virus (YFV), dengue virus (DENV), and Zika virus (ZKV), all of which are global public health concerns. Although the related flaviviruses hepatitis C virus and human pegivirus (formerly named GBV-C) interfere with T-cell receptor (TCR) signaling by novel RNA and protein-based mechanisms, the effect of other flaviviruses on TCR signaling is unknown. Here, we studied the effect of YFV, DENV, and ZKV on TCR signaling. Both YFV and ZKV replicated in human T cells in vitro; however, only YFV inhibited TCR signaling. This effect was mediated at least in part by the YFV envelope (env) protein coding RNA. Deletion mutagenesis studies demonstrated that expression of a short, YFV env RNA motif (vsRNA) was required and sufficient to inhibit TCR signaling. Expression of this vsRNA and YFV infection of T cells reduced the expression of a Src-kinase regulatory phosphatase (PTPRE), while ZKV infection did not. YFV infection in mice resulted in impaired TCR signaling and PTPRE expression, with associated reduction in murine response to experimental ovalbumin vaccination. Together, these data suggest that viruses within the flavivirus genus inhibit TCR signaling in a species-dependent manner.
Keywords: PTPRE; T-cell receptor; flavivirus; virus-derived short RNA; yellow fever virus.
© The Author 2017. Published by Oxford University Press for the Infectious Diseases Society of America. All rights reserved. For permissions, e-mail: journals.permissions@oup.com.
Figures




Similar articles
-
Detection of anti-premembrane antibody as a specific marker of four flavivirus serocomplexes and its application to serosurveillance in endemic regions.Emerg Microbes Infect. 2024 Dec;13(1):2301666. doi: 10.1080/22221751.2023.2301666. Epub 2024 Jan 22. Emerg Microbes Infect. 2024. PMID: 38163752 Free PMC article.
-
Hepatitis C virus infection inhibits a Src-kinase regulatory phosphatase and reduces T cell activation in vivo.PLoS Pathog. 2017 Feb 24;13(2):e1006232. doi: 10.1371/journal.ppat.1006232. eCollection 2017 Feb. PLoS Pathog. 2017. PMID: 28235043 Free PMC article.
-
Characterization of "Off-Target" Immune Modulation Induced by Live Attenuated Yellow Fever Vaccine.J Infect Dis. 2024 Mar 14;229(3):786-794. doi: 10.1093/infdis/jiad086. J Infect Dis. 2024. PMID: 36994927 Free PMC article.
-
Hepatic damage caused by flaviviruses: A systematic review.Life Sci. 2023 Oct 15;331:122074. doi: 10.1016/j.lfs.2023.122074. Epub 2023 Sep 6. Life Sci. 2023. PMID: 37683724
-
Signs and symptoms to determine if a patient presenting in primary care or hospital outpatient settings has COVID-19.Cochrane Database Syst Rev. 2022 May 20;5(5):CD013665. doi: 10.1002/14651858.CD013665.pub3. Cochrane Database Syst Rev. 2022. PMID: 35593186 Free PMC article.
Cited by
-
What Does the Future Hold for Yellow Fever Virus? (II).Genes (Basel). 2018 Aug 21;9(9):425. doi: 10.3390/genes9090425. Genes (Basel). 2018. PMID: 30134625 Free PMC article. Review.
-
Glycerol Monolaurate, an Analogue to a Factor Secreted by Lactobacillus, Is Virucidal against Enveloped Viruses, Including HIV-1.mBio. 2020 May 5;11(3):e00686-20. doi: 10.1128/mBio.00686-20. mBio. 2020. PMID: 32371599 Free PMC article.
-
Flow Cytometry as Immunoassay Tool for Research on Yellow Fever Virus.Methods Mol Biol. 2025;2913:1-17. doi: 10.1007/978-1-0716-4458-4_1. Methods Mol Biol. 2025. PMID: 40249420
-
T-Cell Expression of Angiotensin-Converting Enzyme 2 and Binding of Severe Acute Respiratory Coronavirus 2.J Infect Dis. 2022 Mar 2;225(5):810-819. doi: 10.1093/infdis/jiab595. J Infect Dis. 2022. PMID: 34918095 Free PMC article.
-
Inactivation of Severe Acute Respiratory Coronavirus Virus 2 (SARS-CoV-2) and Diverse RNA and DNA Viruses on Three-Dimensionally Printed Surgical Mask Materials.Infect Control Hosp Epidemiol. 2021 Mar;42(3):253-260. doi: 10.1017/ice.2020.417. Epub 2020 Aug 12. Infect Control Hosp Epidemiol. 2021. PMID: 32783787 Free PMC article.
References
-
- Medzhitov R, Janeway CA Jr. Innate immune recognition and control of adaptive immune responses. Semin Immunol 1998; 10:351–3. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

