Alternative technique or mitigating strategy for sevoflurane-induced neurodegeneration: a randomized controlled dose-escalation study of dexmedetomidine in neonatal rats
- PMID: 28969315
- PMCID: PMC6279114
- DOI: 10.1093/bja/aex219
Alternative technique or mitigating strategy for sevoflurane-induced neurodegeneration: a randomized controlled dose-escalation study of dexmedetomidine in neonatal rats
Abstract
Background: Brain injury in newborn animals from prolonged anaesthetic exposure has raised concerns for millions of children undergoing anaesthesia every yr. Alternative anaesthetic techniques or mitigating strategies are urgently needed to ameliorate potentially harmful effects. We tested dexmedetomidine, both as a single agent alternative technique and as a mitigating adjuvant for sevoflurane anaesthesia.
Methods: Neonatal rats were randomized to three injections of dexmedetomidine (5, 25, 50, or 100 µg kg -1 every 2 h), or 6 h of 2.5% sevoflurane as a single agent without or with dexmedetomidine (1, 5, 10, or 20 µg kg -1 every 2 h). Heart rate, oxygen saturation, level of consciousness, and response to pain were assessed. Cell death was quantified in several brain regions.
Results: Dexmedetomidine provided lower levels of sedation and pain control than sevoflurane. Exposure to either sevoflurane or dexmedetomidine alone did not cause mortality, but the combination of 2.5% sevoflurane and dexmedetomidine in doses exceeding 1 µg kg -1 did. Sevoflurane increased apoptosis in all brain regions; supplementation with dexmedetomidine exacerbated neuronal injury, potentially as a result of ventilatory or haemodynamic compromise. Dexmedetomidine by itself increased apoptosis only in CA2/3 and the ventral posterior nucleus, but not in prefrontal cortex, retrosplenial cortex, somatosensory cortex, subiculum, lateral dorsal thalamic nucleaus, or hippocampal CA1.
Conclusions: We confirm previous findings of sevoflurane-induced neuronal injury. Dexmedetomidine, even in the highest dose, did not cause similar injury, but provided lesser degrees of anaesthesia and pain control. No mitigation of sevoflurane-induced injury was observed with dexmedetomidine supplementation, suggesting that future studies should focus on anaesthetic-sparing effects of dexmedetomidine, rather than injury-preventing effects.
Keywords: anaesthetics; apoptosis; brain injury; dexmedetomidine; inhalation; neuroprotection; safety; sevoflurane; toxicity.
© The Author 2017. Published by Oxford University Press on behalf of the British Journal of Anaesthesia. All rights reserved. For Permissions, please email: journals.permissions@oup.com
Figures
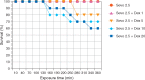
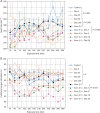



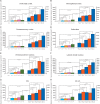
Comment in
-
Reproducibility of science and developmental anaesthesia neurotoxicity: a tale of two cities.Br J Anaesth. 2017 Sep 1;119(3):451-452. doi: 10.1093/bja/aex221. Br J Anaesth. 2017. PMID: 28969307 No abstract available.
-
Theseus, the Labyrinth, and the Minotaur of anaesthetic-induced developmental neurotoxicity.Br J Anaesth. 2017 Sep 1;119(3):453-455. doi: 10.1093/bja/aex235. Br J Anaesth. 2017. PMID: 28969325 No abstract available.
Similar articles
-
Effect of dexmedetomidine on sevoflurane-induced neurodegeneration in neonatal rats.Br J Anaesth. 2021 May;126(5):1009-1021. doi: 10.1016/j.bja.2021.01.033. Epub 2021 Mar 12. Br J Anaesth. 2021. PMID: 33722372
-
Dexmedetomidine-mediated neuroprotection against sevoflurane-induced neurotoxicity extends to several brain regions in neonatal rats.Br J Anaesth. 2017 Sep 1;119(3):506-516. doi: 10.1093/bja/aex222. Br J Anaesth. 2017. PMID: 28969317
-
Dexmedetomidine reduced sevoflurane-induced neurodegeneration and long-term memory deficits in neonatal rats.Int J Dev Neurosci. 2019 Jun;75:19-26. doi: 10.1016/j.ijdevneu.2019.04.002. Epub 2019 Apr 5. Int J Dev Neurosci. 2019. PMID: 30959098
-
Effect of dexmedetomidine in children undergoing general anesthesia with sevoflurane: a meta-analysis.Braz J Anesthesiol. 2017 Mar-Apr;67(2):193-198. doi: 10.1016/j.bjane.2016.02.007. Epub 2016 Nov 25. Braz J Anesthesiol. 2017. PMID: 28236868 Review.
-
Effect of Dexmedetomidine in children undergoing general anaesthesia with sevoflurane: a meta-analysis and systematic review.J Int Med Res. 2020 Jun;48(6):300060520927530. doi: 10.1177/0300060520927530. J Int Med Res. 2020. PMID: 32583698 Free PMC article.
Cited by
-
Anesthesia and Sedation Exposure and Neurodevelopmental Outcomes in Infants Undergoing Congenital Cardiac Surgery: A Retrospective Cohort Study.Anesthesiology. 2023 Oct 1;139(4):393-404. doi: 10.1097/ALN.0000000000004684. Anesthesiology. 2023. PMID: 37440275 Free PMC article.
-
High-Dose Dexmedetomidine Promotes Apoptosis in Fetal Rat Hippocampal Neurons.Drug Des Devel Ther. 2021 Jun 8;15:2433-2444. doi: 10.2147/DDDT.S300247. eCollection 2021. Drug Des Devel Ther. 2021. PMID: 34135573 Free PMC article.
-
Sevoflurane addiction due to workplace exposure: A case report and literature review.Medicine (Baltimore). 2018 Sep;97(38):e12454. doi: 10.1097/MD.0000000000012454. Medicine (Baltimore). 2018. PMID: 30235733 Free PMC article. Review.
-
Does pediatric anesthesia cause brain damage? - Addressing parental and provider concerns in light of compelling animal studies and seemingly ambivalent human data.Korean J Anesthesiol. 2018 Aug;71(4):255-273. doi: 10.4097/kja.d.18.00165. Epub 2018 Jul 4. Korean J Anesthesiol. 2018. PMID: 29969889 Free PMC article.
-
General Anesthetic-Induced Neurotoxicity in the Immature Brain: Reevaluating the Confounding Factors in the Preclinical Studies.Biomed Res Int. 2020 Jan 7;2020:7380172. doi: 10.1155/2020/7380172. eCollection 2020. Biomed Res Int. 2020. PMID: 31998797 Free PMC article. Review.
References
-
- Lin EP, Lee JR, Lee CS, Deng M, Loepke AW.. Do anesthetics harm the developing human brain? an integrative analysis of animal and human studies. Neurotoxicol Teratol 2017; 60: 117–28 - PubMed
-
- Jevtovic-Todorovic V, Olney JW.. Pro: anesthesia-induced developmental neuroapoptosis: status of the evidence. Anesth Analg 2008; 106: 1659–63 - PubMed
-
- Loepke AW, McGowan FX Jr, Soriano SG.. Con: the toxic effects of anesthetics in the developing brain: the clinical perspective. Anesth Analg 2008; 106: 1664–9 - PubMed
-
- Satomoto M, Satoh Y, Terui K, et al.Neonatal exposure to sevoflurane induces abnormal social behaviors and deficits in fear conditioning in mice. Anesthesiology 2009; 110: 628–37 - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

