Peripheral and orofacial pain sensation is unaffected by the loss of p39
- PMID: 28969475
- PMCID: PMC5656108
- DOI: 10.1177/1744806917737205
Peripheral and orofacial pain sensation is unaffected by the loss of p39
Abstract
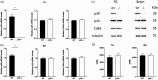
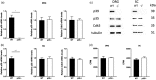
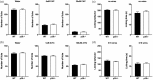
Cdk5 is a key neuronal kinase necessary for proper brain development, which has recently been implicated in modulating nociception. Conditional deletion of Cdk5 in pain-sensing neurons attenuates pain responses to heat in both the periphery and orofacial regions. Cdk5 activity is regulated by binding to the activators p35 and p39, both of which possess a cyclin box. Our previous examination of the nociceptive role of the well-characterized Cdk5 activator p35 using mice that either lack or overexpress this regulatory subunit demonstrated that Cdk5/p35 activity affects mechanical, chemical, and thermal nociception. In contrast, the nociceptive role of Cdk5’s other less-studied activator p39 is unknown. Here, we report that the knockout of p39 in mice did not affect orofacial and peripheral nociception. The lack of any algesic response to nociceptive stimuli in the p39 knockout mice contrasts with the hypoalgesic effects that result from the deletion of p35. Our data demonstrate different and nonoverlapping roles of Cdk5 activators in the regulation of orofacial as well as peripheral nociception with a crucial role for Cdk5/p35 in pain signaling.
Figures
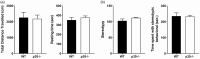
Similar articles
-
p39 Is Responsible for Increasing Cdk5 Activity during Postnatal Neuron Differentiation and Governs Neuronal Network Formation and Epileptic Responses.J Neurosci. 2016 Nov 2;36(44):11283-11294. doi: 10.1523/JNEUROSCI.1155-16.2016. J Neurosci. 2016. PMID: 27807169 Free PMC article.
-
p39, the primary activator for cyclin-dependent kinase 5 (Cdk5) in oligodendroglia, is essential for oligodendroglia differentiation and myelin repair.J Biol Chem. 2013 Jun 21;288(25):18047-57. doi: 10.1074/jbc.M113.453688. Epub 2013 May 3. J Biol Chem. 2013. PMID: 23645679 Free PMC article.
-
Activation of cyclin-dependent kinase 5 mediates orofacial mechanical hyperalgesia.Mol Pain. 2013 Dec 21;9:66. doi: 10.1186/1744-8069-9-66. Mol Pain. 2013. PMID: 24359609 Free PMC article.
-
The regulation of cyclin-dependent kinase 5 activity through the metabolism of p35 or p39 Cdk5 activator.Neurosignals. 2003 Sep-Oct;12(4-5):221-9. doi: 10.1159/000074624. Neurosignals. 2003. PMID: 14673209 Review.
-
Cdk5: a new player in pain signaling.Cell Cycle. 2006 Mar;5(6):585-8. doi: 10.4161/cc.5.6.2578. Epub 2006 Mar 15. Cell Cycle. 2006. PMID: 16552189 Review.
Cited by
-
Valproic Acid-Induced Anxiety and Depression Behaviors are Ameliorated in p39 Cdk5 Activator-Deficient Mice.Neurochem Res. 2022 Sep;47(9):2773-2779. doi: 10.1007/s11064-022-03642-9. Epub 2022 Jun 8. Neurochem Res. 2022. PMID: 35674931
-
Nociceptive signaling through transient receptor potential vanilloid 1 is regulated by Cyclin Dependent Kinase 5-mediated phosphorylation of T407 in vivo.Mol Pain. 2022 Apr;18:17448069221111473. doi: 10.1177/17448069221111473. Mol Pain. 2022. PMID: 35726573 Free PMC article.
-
Specific 3-O-sulfated heparan sulfate domains regulate salivary gland basement membrane metabolism and epithelial differentiation.Nat Commun. 2024 Aug 31;15(1):7584. doi: 10.1038/s41467-024-51862-0. Nat Commun. 2024. PMID: 39217171 Free PMC article.
-
Loss of Hs3st3a1 or Hs3st3b1 enzymes alters heparan sulfate to reduce epithelial morphogenesis and adult salivary gland function.Matrix Biol. 2021 Sep;103-104:37-57. doi: 10.1016/j.matbio.2021.10.002. Epub 2021 Oct 12. Matrix Biol. 2021. PMID: 34653670 Free PMC article.
-
Protocols for Characterization of Cdk5 Kinase Activity.Curr Protoc. 2021 Oct;1(10):e276. doi: 10.1002/cpz1.276. Curr Protoc. 2021. PMID: 34679246 Free PMC article.
References
-
- Su SC, Tsai LH. Cyclin-dependent kinases in brain development and disease. Annu Rev Cell Dev Biol 2011; 27: 465–491. - PubMed
-
- Dhavan R, Tsai LH. A decade of CDK5. Nat Rev Mol Cell Biol 2001; 2: 749–759. - PubMed
-
- Lalioti V, Pulido D, Sandoval IV. Cdk5, the multifunctional surveyor. Cell Cycle 2010; 9: 284–311. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

