Imaging the polymerization of multivalent nanoparticles in solution
- PMID: 28970557
- PMCID: PMC5624893
- DOI: 10.1038/s41467-017-00857-1
Imaging the polymerization of multivalent nanoparticles in solution
Abstract
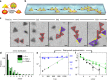
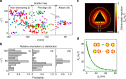
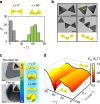
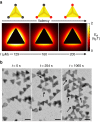
Numerous mechanisms have been studied for chemical reactions to provide quantitative predictions on how atoms spatially arrange into molecules. In nanoscale colloidal systems, however, less is known about the physical rules governing their spatial organization, i.e., self-assembly, into functional materials. Here, we monitor real-time self-assembly dynamics at the single nanoparticle level, which reveal marked similarities to foundational principles of polymerization. Specifically, using the prototypical system of gold triangular nanoprisms, we show that colloidal self-assembly is analogous to polymerization in three aspects: ensemble growth statistics following models for step-growth polymerization, with nanoparticles as linkable "monomers"; bond angles determined by directional internanoparticle interactions; and product topology determined by the valency of monomeric units. Liquid-phase transmission electron microscopy imaging and theoretical modeling elucidate the nanometer-scale mechanisms for these polymer-like phenomena in nanoparticle systems. The results establish a quantitative conceptual framework for self-assembly dynamics that can aid in designing future nanoparticle-based materials.Few models exist that describe the spontaneous organization of colloids into materials. Here, the authors combine liquid-phase TEM and single particle tracking to observe the dynamics of gold nanoprisms, finding that nanoscale self-assembly can be understood within the framework of atomic polymerization.
Conflict of interest statement
The authors declare no competing financial interests.
Figures

Similar articles
-
Quantifying the Self-Assembly Behavior of Anisotropic Nanoparticles Using Liquid-Phase Transmission Electron Microscopy.Acc Chem Res. 2017 May 16;50(5):1125-1133. doi: 10.1021/acs.accounts.7b00048. Epub 2017 Apr 26. Acc Chem Res. 2017. PMID: 28443654
-
Step-growth polymerization of inorganic nanoparticles.Science. 2010 Jul 9;329(5988):197-200. doi: 10.1126/science.1189457. Science. 2010. PMID: 20616274 Free PMC article.
-
Visualization of Colloidal Nanocrystal Formation and Electrode-Electrolyte Interfaces in Liquids Using TEM.Acc Chem Res. 2017 Aug 15;50(8):1808-1817. doi: 10.1021/acs.accounts.7b00161. Epub 2017 Aug 7. Acc Chem Res. 2017. PMID: 28782932
-
Liquid-Phase Transmission Electron Microscopy for Studying Colloidal Inorganic Nanoparticles.Adv Mater. 2018 Jan;30(4). doi: 10.1002/adma.201703316. Epub 2017 Nov 27. Adv Mater. 2018. PMID: 29178589 Review.
-
Colloidal gold and silver triangular nanoprisms.Small. 2009 Mar;5(6):646-64. doi: 10.1002/smll.200801480. Small. 2009. PMID: 19306458 Review.
Cited by
-
Hierarchical self-assembly of 3D lattices from polydisperse anisometric colloids.Nat Commun. 2019 Apr 18;10(1):1815. doi: 10.1038/s41467-019-09787-6. Nat Commun. 2019. PMID: 31000717 Free PMC article.
-
Intracellular Thermal Probing Using Aggregated Fluorescent Nanodiamonds.Adv Sci (Weinh). 2022 Jan;9(3):e2103354. doi: 10.1002/advs.202103354. Epub 2021 Nov 23. Adv Sci (Weinh). 2022. PMID: 34813176 Free PMC article.
-
Exploiting limited valence patchy particles to understand autocatalytic kinetics.Nat Commun. 2018 Jul 6;9(1):2647. doi: 10.1038/s41467-018-04977-0. Nat Commun. 2018. PMID: 29980675 Free PMC article.
-
Machine Learning to Reveal Nanoparticle Dynamics from Liquid-Phase TEM Videos.ACS Cent Sci. 2020 Aug 26;6(8):1421-1430. doi: 10.1021/acscentsci.0c00430. Epub 2020 Jul 6. ACS Cent Sci. 2020. PMID: 32875083 Free PMC article.
-
Formation of self-assembled gold nanoparticle supercrystals with facet-dependent surface plasmonic coupling.Nat Commun. 2018 Jun 18;9(1):2365. doi: 10.1038/s41467-018-04801-9. Nat Commun. 2018. PMID: 29915321 Free PMC article.
References
-
- De Yoreo JJ, Vekilov PG. Principles of crystal nucleation and growth. Rev. Miner. Geochem. 2003;54:57–93. doi: 10.2113/0540057. - DOI
-
- Anslyn, E. V. & Dougherty, D. A. Modern Physical Organic Chemistry. (University Science Books, 2005).
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources

