Effect of Inhaled Nitric Oxide on Survival Without Bronchopulmonary Dysplasia in Preterm Infants: A Randomized Clinical Trial
- PMID: 28973344
- PMCID: PMC5710365
- DOI: 10.1001/jamapediatrics.2017.2618
Effect of Inhaled Nitric Oxide on Survival Without Bronchopulmonary Dysplasia in Preterm Infants: A Randomized Clinical Trial
Abstract
Importance: Bronchopulmonary dysplasia (BPD) occurs in approximately 40% of infants born at younger than 30 weeks' gestation and is associated with adverse pulmonary and neurodevelopmental outcomes.
Objective: To test whether administration of inhaled nitric oxide to preterm infants requiring positive pressure respiratory support on postnatal days 5 to 14 improves the rate of survival without BPD.
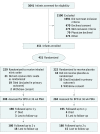
Design, setting, and participants: This intent-to-treat study was a randomized clinical trial performed at 33 US and Canadian neonatal intensive care units. Participants included 451 neonates younger than 30 weeks' gestation with birth weight less than 1250 g receiving mechanical ventilation or positive pressure respiratory support on postnatal days 5 to 14. Enrollment spanned from December 23, 2009, to April 23, 2012, and neurodevelopmental outcome studies were completed by April 4, 2014.
Interventions: Placebo (nitrogen) or inhaled nitric oxide initiated at 20 ppm was decreased to 10 ppm between 72 and 96 hours after starting treatment and then to 5 ppm on day 10 or 11. Infants remained on the 5-ppm dose until completion of therapy (24 days).
Main outcomes and measures: The primary outcome was the rate of survival without BPD at 36 weeks' postmenstrual age (PMA). Secondary outcomes included BPD severity, postnatal corticosteroid use, respiratory support, survival, and neurodevelopmental outcomes at 18 to 24 months' PMA.
Results: In total, 222 infants (52.3% male [n = 116]) received placebo, and 229 infants (50.2% male [n = 115]) received inhaled nitric oxide. Their mean (SD) gestation was 25.6 (1.5) vs 25.6 (1.4) weeks, and their mean (SD) birth weight was 750 (164) vs 724 (160) g. Survival without BPD at 36 weeks' PMA was similar between the placebo and inhaled nitric oxide groups (31.5% [n = 70] vs 34.9% [n = 80]) (odds ratio, 1.17; 95% CI, 0.79-1.73). Rates for severe BPD (26.6% [55 of 207] vs 20.5% [43 of 210]) and postnatal corticosteroid use for BPD (41.0% [91 of 222] vs 41.5% [95 of 229]) and the mean (SD) days of positive pressure respiratory support (55 [40] vs 54 [42]), oxygen therapy (88 [41] vs 91 [59]), and hospitalization (105 [37] vs 108 [54]) were equivalent between the 2 groups. No differences in the incidence of common morbidities were observed. Respiratory outcomes on discharge to home, at 1 year, and at age 18 to 24 months' PMA and neurodevelopmental assessments at 18 to 24 months' PMA did not differ between groups.
Conclusions and relevance: Inhaled nitric oxide, initiated at 20 ppm on postnatal days 5 to 14 to high-risk preterm infants and continued for 24 days, appears to be safe but did not improve survival without BPD at 36 weeks' PMA or respiratory and neurodevelopmental outcomes at 18 to 24 months' PMA.
Trial registration: clinicaltrials.gov Identifier: NCT00931632.
Conflict of interest statement
Figures
Comment in
-
The death knell of inhaled nitric oxide to prevent bronchopulmonary dysplasia?J Perinatol. 2018 Jun;38(6):633-635. doi: 10.1038/s41372-018-0071-5. Epub 2018 Feb 21. J Perinatol. 2018. PMID: 29467517 No abstract available.
References
-
- Vrijlandt EJ, Boezen HM, Gerritsen J, Stremmelaar EF, Duiverman EJ. Respiratory health in prematurely born preschool children with and without bronchopulmonary dysplasia. J Pediatr. 2007;150(3):256-261. - PubMed
-
- Northway WH Jr, Moss RB, Carlisle KB, et al. Late pulmonary sequelae of bronchopulmonary dysplasia. N Engl J Med. 1990;323(26):1793-1799. - PubMed
-
- Wood NS, Costeloe K, Gibson AT, Hennessy EM, Marlow N, Wilkinson AR; EPICure Study Group . The EPICure study: associations and antecedents of neurological and developmental disability at 30 months of age following extremely preterm birth. Arch Dis Child Fetal Neonatal Ed. 2005;90(2):F134-F140. - PMC - PubMed
-
- Vohr BR, Wright LL, Poole WK, McDonald SA. Neurodevelopmental outcomes of extremely low birth weight infants <32 weeks’ gestation between 1993 and 1998. Pediatrics. 2005;116(3):635-643. - PubMed
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous