Don't let sleeping dogmas lie: new views of peptidoglycan synthesis and its regulation
- PMID: 28975672
- PMCID: PMC5720918
- DOI: 10.1111/mmi.13853
Don't let sleeping dogmas lie: new views of peptidoglycan synthesis and its regulation
Abstract
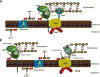
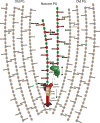
Bacterial cell wall synthesis is the target for some of our most powerful antibiotics and has thus been the subject of intense research focus for more than 50 years. Surprisingly, we still lack a fundamental understanding of how bacteria build, maintain and expand their cell wall. Due to technical limitations, directly testing hypotheses about the coordination and biochemistry of cell wall synthesis enzymes or architecture has been challenging, and interpretation of data has therefore often relied on circumstantial evidence and implicit assumptions. A number of recent papers have exploited new technologies, like single molecule tracking and real-time, high resolution temporal mapping of cell wall synthesis processes, to address fundamental questions of bacterial cell wall biogenesis. The results have challenged established dogmas and it is therefore timely to integrate new data and old observations into a new model of cell wall biogenesis in rod-shaped bacteria.
© 2017 John Wiley & Sons Ltd.
Figures
References
-
- Anderson JS, Matsuhashi M, Haskin MA, Strominger JL. Biosythesis of the peptidoglycan of bacterial cell walls. II. Phospholipid carriers in the reaction sequence. J Biol Chem. 1967;242:3180–3190. - PubMed
-
- Anderson JS, Meadow PM, Haskin MA, Strominger JL. Biosynthesis of the peptidoglycan of bacterial cell walls. I. Utilization of uridine diphosphate acetylmuramyl pentapeptide and uridine diphosphate acetylglucosamine for peptidoglycan synthesis by particulate enzymes from Staphylococcus aureus and Micrococcus lysodeikticus. Arch Biochem Biophys. 1966;116:487–515. - PubMed
-
- Banzhaf M, van den Berg van Saparoea B, Terrak M, Fraipont C, Egan A, Philippe J, Zapun A, Breukink E, Nguyen-Disteche M, den Blaauwen T, Vollmer W. Cooperativity of peptidoglycan synthases active in bacterial cell elongation. Mol Microbiol. 2012;85:179–194. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

