Acquisition of the T790M resistance mutation during afatinib treatment in EGFR tyrosine kinase inhibitor-naïve patients with non-small cell lung cancer harboring EGFR mutations
- PMID: 28978102
- PMCID: PMC5620242
- DOI: 10.18632/oncotarget.19243
Acquisition of the T790M resistance mutation during afatinib treatment in EGFR tyrosine kinase inhibitor-naïve patients with non-small cell lung cancer harboring EGFR mutations
Abstract
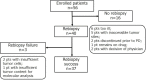
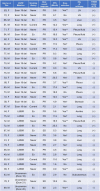
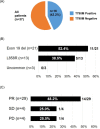
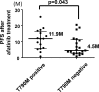
The T790M secondary mutation of the epidermal growth factor receptor (EGFR) gene accounts for 50% to 60% of cases of resistance to the first-generation EGFR tyrosine kinase inhibitors (TKIs) gefitinib and erlotinib. The prevalence of T790M in EGFR mutation-positive patients who acquire resistance to the irreversible, second-generation EGFR-TKI afatinib has remained unclear, however. We here determined the frequency of T790M acquisition at diagnosis of progressive disease in patients with EGFR-mutated non-small cell lung cancer (NSCLC) treated with afatinib as first-line EGFR-TKI. Among 56 enrolled patients, 37 individuals underwent molecular analysis at rebiopsy. Of these 37 patients, 16 individuals (43.2%) had acquired T790M, including 11/21 patients (52.4%) with an exon 19 deletion of EGFR and 5/13 patients (38.5%) with L858R. None of three patients with an uncommon EGFR mutation harbored T790M. T790M was detected in 14/29 patients (48.3%) with a partial response to afatinib, 1/4 patients (25%) with stable disease, and 1/4 patients (25%) with progressive disease as the best response. Median progression-free survival after initiation of afatinib treatment was significantly (P = 0.043) longer in patients who acquired T790M (11.9 months; 95% confidence interval, 8.7-15.1) than in those who did not (4.5 months; 95% confidence interval, 2.0-7.0). Together, our results show that EGFR-mutated NSCLC patients treated with afatinib as first-line EGFR-TKI acquire T790M at the time of progression at a frequency similar to that for patients treated with gefitinib or erlotinib. They further underline the importance of rebiopsy for detection of T790M in afatinib-treated patients.
Keywords: T790M; acquired resistance; afatinib; non–small cell lung cancer (NSCLC); rebiopsy.
Conflict of interest statement
CONFLICTS OF INTERESTS Kentaro Tanaka has received honoraria from Nippon Boehringer Ingelheim. Kaname Nosaki has received research funding from MSD and Novartis Pharma as well as honoraria from AstraZeneca, Chugai, Eli Lilly, Kyowa Hakko Kirin, Nippon Boehringer Ingelheim, Nippon Kayaku, ONO, and MSD. Kohei Otsubo has received honoraria from Nippon Boehringer Ingelheim. Hiroshi Wataya has received honoraria from Nippon Boehringer Ingelheim. Taishi Harada has received honoraria from Nippon Boehringer Ingelheim. Yoichi Nakanishi has received research funding and honoraria from Nippon Boehringer Ingelheim. Isamu Okamoto has received research funding and honoraria from Nippon Boehringer Ingelheim.
Figures
Similar articles
-
An Observational Study of Acquired EGFR T790M-Dependent Resistance to EGFR-TKI Treatment in Lung Adenocarcinoma Patients in Taiwan.Front Oncol. 2020 Sep 4;10:1481. doi: 10.3389/fonc.2020.01481. eCollection 2020. Front Oncol. 2020. PMID: 33014788 Free PMC article.
-
Activity of the EGFR-HER2 dual inhibitor afatinib in EGFR-mutant lung cancer patients with acquired resistance to reversible EGFR tyrosine kinase inhibitors.Clin Lung Cancer. 2014 Nov;15(6):411-417.e4. doi: 10.1016/j.cllc.2014.07.002. Epub 2014 Aug 16. Clin Lung Cancer. 2014. PMID: 25242668
-
Afatinib overcoming resistance to icotinib and osimertinib in NSCLC with leptomeningeal metastasis in patients with acquired EGFR L858R/T790M or L858R/S768I mutations: Two case reports.Heliyon. 2023 Oct 8;9(10):e20690. doi: 10.1016/j.heliyon.2023.e20690. eCollection 2023 Oct. Heliyon. 2023. PMID: 37860534 Free PMC article.
-
Dacomitinib in lung cancer: a "lost generation" EGFR tyrosine-kinase inhibitor from a bygone era?Drug Des Devel Ther. 2015 Oct 15;9:5641-53. doi: 10.2147/DDDT.S52787. eCollection 2015. Drug Des Devel Ther. 2015. PMID: 26508839 Free PMC article. Review.
-
Comparison of T790M Acquisition After Treatment With First- and Second-Generation Tyrosine-Kinase Inhibitors: A Systematic Review and Network Meta-Analysis.Front Oncol. 2022 Jun 28;12:869390. doi: 10.3389/fonc.2022.869390. eCollection 2022. Front Oncol. 2022. PMID: 35837103 Free PMC article.
Cited by
-
Absence of copy number gain of EGFR: A possible predictive marker of long-term response to afatinib.Cancer Sci. 2023 Mar;114(3):1045-1055. doi: 10.1111/cas.15655. Epub 2022 Dec 16. Cancer Sci. 2023. PMID: 36382532 Free PMC article.
-
Exploration of resistance mechanisms for epidermal growth factor receptor-tyrosine kinase inhibitors based on plasma analysis by digital polymerase chain reaction and next-generation sequencing.Cancer Sci. 2018 Dec;109(12):3921-3933. doi: 10.1111/cas.13820. Epub 2018 Nov 13. Cancer Sci. 2018. PMID: 30289575 Free PMC article.
-
Afatinib as First-Line Treatment in Asian Patients with EGFR Mutation-Positive NSCLC: A Narrative Review of Real-World Evidence.Adv Ther. 2021 May;38(5):2038-2053. doi: 10.1007/s12325-021-01696-9. Epub 2021 Mar 17. Adv Ther. 2021. PMID: 33730350 Free PMC article. Review.
-
Liquid-Biopsy-Based Identification of EGFR T790M Mutation-Mediated Resistance to Afatinib Treatment in Patients with Advanced EGFR Mutation-Positive NSCLC, and Subsequent Response to Osimertinib.Target Oncol. 2019 Feb;14(1):75-83. doi: 10.1007/s11523-018-0612-z. Target Oncol. 2019. PMID: 30539501 Free PMC article. Clinical Trial.
-
A Multicenter Study to Assess EGFR Mutational Status in Plasma: Focus on an Optimized Workflow for Liquid Biopsy in a Clinical Setting.Cancers (Basel). 2018 Aug 27;10(9):290. doi: 10.3390/cancers10090290. Cancers (Basel). 2018. PMID: 30150518 Free PMC article.
References
-
- Mok TS, Wu YL, Thongprasert S, Yang CH, Chu DT, Saijo N, Sunpaweravong P, Han B, Margono B, Ichinose Y, Nishiwaki Y, Ohe Y, Yang JJ, et al. Gefitinib or carboplatin-paclitaxel in pulmonary adenocarcinoma. New Engl J Med. 2009;361:947–57. - PubMed
-
- Mitsudomi T, Morita S, Yatabe Y, Negoro S, Okamoto I, Tsurutani J, Seto T, Satouchi M, Tada H, Hirashima T, Asami K, Katakami N, Takada M, et al. Gefitinib versus cisplatin plus docetaxel in patients with non-small-cell lung cancer harbouring mutations of the epidermal growth factor receptor (WJTOG3405): an open label, randomised phase 3 trial. Lancet Oncol. 2010;11:121–8. - PubMed
-
- Maemondo M, Inoue A, Kobayashi K, Sugawara S, Oizumi S, Isobe H, Gemma A, Harada M, Yoshizawa H, Kinoshita I, Fujita Y, Okinaga S, Hirano H, et al. Gefitinib or chemotherapy for non-small-cell lung cancer with mutated EGFR. New Engl J Med. 2010;362:2380–8. - PubMed
-
- Sequist LV, Waltman BA, Dias-Santagata D, Digumarthy S, Turke AB, Fidias P, Bergethon K, Shaw AT, Gettinger S, Cosper AK, Akhavanfard S, Heist RS, Temel J, et al. Genotypic and histological evolution of lung cancers acquiring resistance to EGFR inhibitors. Sci Transl Med. 2011;3:75ra26. - PMC - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

