Quantitative Deformability Cytometry: Rapid, Calibrated Measurements of Cell Mechanical Properties
- PMID: 28978449
- PMCID: PMC5627151
- DOI: 10.1016/j.bpj.2017.06.073
Quantitative Deformability Cytometry: Rapid, Calibrated Measurements of Cell Mechanical Properties
Abstract
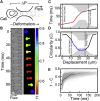
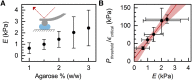
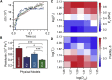
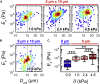
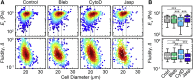
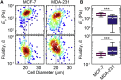
Advances in methods that determine cell mechanical phenotype, or mechanotype, have demonstrated the utility of biophysical markers in clinical and research applications ranging from cancer diagnosis to stem cell enrichment. Here, we introduce quantitative deformability cytometry (q-DC), a method for rapid, calibrated, single-cell mechanotyping. We track changes in cell shape as cells deform into microfluidic constrictions, and we calibrate the mechanical stresses using gel beads. We observe that time-dependent strain follows power-law rheology, enabling single-cell measurements of apparent elastic modulus, Ea, and power-law exponent, β. To validate our method, we mechanotype human promyelocytic leukemia (HL-60) cells and thereby confirm q-DC measurements of Ea = 0.53 ± 0.04 kPa. We also demonstrate that q-DC is sensitive to pharmacological perturbations of the cytoskeleton as well as differences in the mechanotype of human breast cancer cell lines (Ea = 2.1 ± 0.1 and 0.80 ± 0.19 kPa for MCF-7 and MDA-MB-231 cells). To establish an operational framework for q-DC, we investigate the effects of applied stress and cell/pore-size ratio on mechanotype measurements. We show that Ea increases with applied stress, which is consistent with stress stiffening behavior of cells. We also find that Ea increases for larger cell/pore-size ratios, even when the same applied stress is maintained; these results indicate strain stiffening and/or dependence of mechanotype on deformation depth. Taken together, the calibrated measurements enabled by q-DC should advance applications of cell mechanotype in basic research and clinical settings.
Copyright © 2017 Biophysical Society. Published by Elsevier Inc. All rights reserved.
Figures
Similar articles
-
The physical origins of transit time measurements for rapid, single cell mechanotyping.Lab Chip. 2016 Aug 16;16(17):3330-9. doi: 10.1039/c6lc00169f. Lab Chip. 2016. PMID: 27435631
-
Microfluidic Mechanotyping of a Single Cell with Two Consecutive Constrictions of Different Sizes and an Electrical Detection System.Anal Chem. 2019 Oct 15;91(20):12890-12899. doi: 10.1021/acs.analchem.9b02818. Epub 2019 Sep 11. Anal Chem. 2019. PMID: 31442026
-
Microconstriction arrays for high-throughput quantitative measurements of cell mechanical properties.Biophys J. 2015 Jul 7;109(1):26-34. doi: 10.1016/j.bpj.2015.05.029. Biophys J. 2015. PMID: 26153699 Free PMC article.
-
Microfluidic characterization of single-cell biophysical properties and the applications in cancer diagnosis.Electrophoresis. 2024 Jul;45(13-14):1212-1232. doi: 10.1002/elps.202300177. Epub 2023 Nov 1. Electrophoresis. 2024. PMID: 37909658 Review.
-
Recent advances in microfluidic techniques for single-cell biophysical characterization.Lab Chip. 2013 Jul 7;13(13):2464-83. doi: 10.1039/c3lc50355k. Epub 2013 May 16. Lab Chip. 2013. PMID: 23681312 Review.
Cited by
-
Acousto-holographic reconstruction of whole-cell stiffness maps.Nat Commun. 2022 Nov 29;13(1):7351. doi: 10.1038/s41467-022-35075-x. Nat Commun. 2022. PMID: 36446776 Free PMC article.
-
Single-cell mechanical analysis and tension quantification via electrodeformation relaxation.Phys Rev E. 2021 Mar;103(3-1):032409. doi: 10.1103/PhysRevE.103.032409. Phys Rev E. 2021. PMID: 33862816 Free PMC article.
-
Elasticity of Carrier Fluid: A Key Factor Affecting Mechanical Phenotyping in Deformability Cytometry.Micromachines (Basel). 2024 Jun 25;15(7):822. doi: 10.3390/mi15070822. Micromachines (Basel). 2024. PMID: 39064333 Free PMC article.
-
Unraveling the mechanobiology of immune cells.Curr Opin Biotechnol. 2020 Dec;66:236-245. doi: 10.1016/j.copbio.2020.09.004. Epub 2020 Sep 30. Curr Opin Biotechnol. 2020. PMID: 33007634 Free PMC article. Review.
-
Biophysical phenotyping of cells via impedance spectroscopy in parallel cyclic deformability channels.Biomicrofluidics. 2019 Jul 18;13(4):044103. doi: 10.1063/1.5099269. eCollection 2019 Jul. Biomicrofluidics. 2019. PMID: 31341524 Free PMC article.
References
-
- Engler A.J., Sen S., Discher D.E. Matrix elasticity directs stem cell lineage specification. Cell. 2006;126:677–689. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

