Post-transcriptional Inhibition of Hsc70-4/HSPA8 Expression Leads to Synaptic Vesicle Cycling Defects in Multiple Models of ALS
- PMID: 28978466
- PMCID: PMC5679478
- DOI: 10.1016/j.celrep.2017.09.028
Post-transcriptional Inhibition of Hsc70-4/HSPA8 Expression Leads to Synaptic Vesicle Cycling Defects in Multiple Models of ALS
Abstract
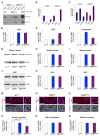
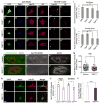
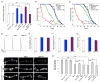
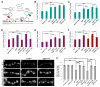
Amyotrophic lateral sclerosis (ALS) is a synaptopathy accompanied by the presence of cytoplasmic aggregates containing TDP-43, an RNA-binding protein linked to ∼97% of ALS cases. Using a Drosophila model of ALS, we show that TDP-43 overexpression (OE) in motor neurons results in decreased expression of the Hsc70-4 chaperone at the neuromuscular junction (NMJ). Mechanistically, mutant TDP-43 sequesters hsc70-4 mRNA and impairs its translation. Expression of the Hsc70-4 ortholog, HSPA8, is also reduced in primary motor neurons and NMJs of mice expressing mutant TDP-43. Electrophysiology, imaging, and genetic interaction experiments reveal TDP-43-dependent defects in synaptic vesicle endocytosis. These deficits can be partially restored by OE of Hsc70-4, cysteine-string protein (Csp), or dynamin. This suggests that TDP-43 toxicity results in part from impaired activity of the synaptic CSP/Hsc70 chaperone complex impacting dynamin function. Finally, Hsc70-4/HSPA8 expression is also post-transcriptionally reduced in fly and human induced pluripotent stem cell (iPSC) C9orf72 models, suggesting a common disease pathomechanism.
Keywords: C9orf72; Drosophila; RNA processing; TDP-43; amyotrophic lateral sclerosis; endocytosis; iPSC; neuromuscular junction; synaptic vesicle cycle; translation.
Copyright © 2017 The Author(s). Published by Elsevier Inc. All rights reserved.
Figures

Similar articles
-
Defects in synaptic transmission at the neuromuscular junction precede motor deficits in a TDP-43Q331K transgenic mouse model of amyotrophic lateral sclerosis.FASEB J. 2018 May;32(5):2676-2689. doi: 10.1096/fj.201700835R. Epub 2018 Jan 2. FASEB J. 2018. PMID: 29295857
-
Motor neurons and glia exhibit specific individualized responses to TDP-43 expression in a Drosophila model of amyotrophic lateral sclerosis.Dis Model Mech. 2013 May;6(3):721-33. doi: 10.1242/dmm.010710. Epub 2013 Feb 1. Dis Model Mech. 2013. PMID: 23471911 Free PMC article.
-
Homeostatic plasticity can be induced and expressed to restore synaptic strength at neuromuscular junctions undergoing ALS-related degeneration.Hum Mol Genet. 2017 Nov 1;26(21):4153-4167. doi: 10.1093/hmg/ddx304. Hum Mol Genet. 2017. PMID: 28973139 Free PMC article.
-
TDP-43 dysregulation and neuromuscular junction disruption in amyotrophic lateral sclerosis.Transl Neurodegener. 2022 Dec 27;11(1):56. doi: 10.1186/s40035-022-00331-z. Transl Neurodegener. 2022. PMID: 36575535 Free PMC article. Review.
-
Synaptic dysfunction and altered excitability in C9ORF72 ALS/FTD.Brain Res. 2018 Aug 15;1693(Pt A):98-108. doi: 10.1016/j.brainres.2018.02.011. Epub 2018 Feb 14. Brain Res. 2018. PMID: 29453960 Free PMC article. Review.
Cited by
-
Insights into C9ORF72-Related ALS/FTD from Drosophila and iPSC Models.Trends Neurosci. 2018 Jul;41(7):457-469. doi: 10.1016/j.tins.2018.04.002. Epub 2018 May 2. Trends Neurosci. 2018. PMID: 29729808 Free PMC article. Review.
-
Homozygous ALS-linked mutations in TARDBP/TDP-43 lead to hypoactivity and synaptic abnormalities in human iPSC-derived motor neurons.iScience. 2024 Feb 9;27(3):109166. doi: 10.1016/j.isci.2024.109166. eCollection 2024 Mar 15. iScience. 2024. PMID: 38433895 Free PMC article.
-
New Partners Identified by Mass Spectrometry Assay Reveal Functions of NCAM2 in Neural Cytoskeleton Organization.Int J Mol Sci. 2021 Jul 9;22(14):7404. doi: 10.3390/ijms22147404. Int J Mol Sci. 2021. PMID: 34299022 Free PMC article.
-
Acetyl-L-carnitine and Amyotrophic Lateral Sclerosis: Current Evidence and Potential use.CNS Neurol Disord Drug Targets. 2024;23(5):588-601. doi: 10.2174/1871527322666230330083757. CNS Neurol Disord Drug Targets. 2024. PMID: 36998125 Review.
-
In vitro methods in autophagy research: Applications in neurodegenerative diseases and mood disorders.Front Mol Neurosci. 2023 Apr 12;16:1168948. doi: 10.3389/fnmol.2023.1168948. eCollection 2023. Front Mol Neurosci. 2023. PMID: 37122628 Free PMC article. Review.
References
-
- Braun JE, Wilbanks SM, Scheller RH. The cysteine string secretory vesicle protein activates Hsc70 ATPase. J Biol Chem. 1996;271:25989–25993. - PubMed
-
- Bronk P, Wenniger JJ, Dawson-Scully K, Guo X, Hong S, Atwood HL, Zinsmaier KE. Drosophila Hsc70-4 is critical for neurotransmitter exocytosis in vivo. Neuron. 2001;30:475–488. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Miscellaneous

