Synthesis of Titanium Dioxide Nanoparticles Using Echinacea purpurea Herba
- PMID: 28979329
- PMCID: PMC5603885
Synthesis of Titanium Dioxide Nanoparticles Using Echinacea purpurea Herba
Abstract
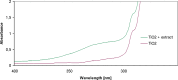
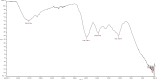
Nowadays green synthesis of metal nanoparticles is a developing area of research. In this study, titanium dioxide nanoparticles were biosynthesized using an aqueous solution of Echinacea purpurea herba extract as a bioreductant. This is novel and interesting method for synthesis of TiO2 nanoparticles. The prepared titanium dioxide nanoparticles were characterized using ultraviolet-visible spectroscopy (UV-VIS), transmission electron microscopy (SEM), total reflection X-Ray fluorescence analysis (TXRF) and Fourier-transform infrared spectroscopy (FTIR). The size of TiO2 nanoparticles was found to be in the range of 120 nm. Moreover, the alkaline reaction of the solution (pH = 8) resulted in the increase in absorbance (280 nm), which facilitates the growth of the number of TiO2 nanoparticles in the studied solution. Also, synthesis of TiO2 nanoparticles using green resources like Echinacea purpurea herba is a better alternative to chemical synthesis, since this green synthesis is pollutant-free and eco-friendly.
Keywords: Echinacea purpurea; green-synthesis; nanotechnology; titanium dioxide nanoparticles.
Figures





References
-
- Ahmed M, Karns M, Goodson M, Rowe J, Hussain SM, Schlager JJ, Hong Y. DNA damage response to different surface chemistry of silver nanoparticles in mammalian cells. Toxicol. Appl. Pharmacol. . 2008;233:404–10. - PubMed
-
- Brayner R. The toxicological impact of nanoparticles. Nanotoday . 2008;3:48–55.
-
- Panda KK, Achary VMM, Krishnaveni R, Padhi BK, Sarangi SN, Sahu SN. In vitro biosynthesis and genotoxicity bioassay of silver nanoparticles using plants. Toxicol. In Vitro . 2011;25:1097–105. - PubMed
-
- Kumar V, Yadav SK. Plant-mediated synthesis of silver and gold nanoparticles and their applications. J. Chem. Technol. Biotechnol. . 2009;84:151–7.
LinkOut - more resources
Full Text Sources
