Active immunotherapy for TNF-mediated inflammation using self-assembled peptide nanofibers
- PMID: 28982051
- PMCID: PMC5716349
- DOI: 10.1016/j.biomaterials.2017.09.031
Active immunotherapy for TNF-mediated inflammation using self-assembled peptide nanofibers
Abstract
Active immunotherapies raising antibody responses against autologous targets are receiving increasing interest as alternatives to the administration of manufactured antibodies. The challenge in such an approach is generating protective and adjustable levels of therapeutic antibodies while at the same time avoiding strong T cell responses that could lead to autoimmune reactions. Here we demonstrate the design of an active immunotherapy against TNF-mediated inflammation using short synthetic peptides that assemble into supramolecular peptide nanofibers. Immunization with these materials, without additional adjuvants, was able to break B cell tolerance and raise protective antibody responses against autologous TNF in mice. The strength of the anti-TNF antibody response could be tuned by adjusting the epitope content in the nanofibers, and the T-cell response was focused on exogenous and non-autoreactive T-cell epitopes. Immunization with unadjuvanted peptide nanofibers was therapeutic in a lethal model of acute inflammation induced by intraperitoneally delivered lipopolysaccharide, whereas formulations adjuvanted with CpG showed comparatively poorer protection that correlated with a more Th1-polarized response. Additionally, immunization with peptide nanofibers did not diminish the ability of mice to clear infections of Listeria monocytogenes. Collectively this work suggests that synthetic self-assembled peptides can be attractive platforms for active immunotherapies against autologous targets.
Keywords: Active immunotherapy; Adjuvant-free; Supramolecular; TNF; Vaccine.
Copyright © 2017. Published by Elsevier Ltd.
Conflict of interest statement
Figures



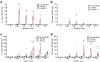

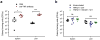
References
-
- Hansel TT, Kropshofer H, Singer T, Mitchell JA, George AJT. The safety and side effects of monoclonal antibodies. Nat Rev Drug Discov. 2010;9:325–38. - PubMed
-
- Ware CF. In: Advances in Pharmacology. Webb DR, editor. Academic Press; 2013. pp. 51–80. - PubMed
-
- Zagury D, Le Buanec H, Bizzini B, Burny A, Lewis G, Gallo RC. Active versus passive anti-cytokine antibody therapy against cytokine-associated chronic diseases. Cytokine Growth Factor Rev. 2003;14:123–37. - PubMed
-
- Jia T, Pan Y, Li J, Wang L. Strategies for active TNF-alpha vaccination in rheumatoid arthritis treatment. Vaccine. 2013;31:4063–8. - PubMed
-
- Dalum I, Butler DM, Jensen MR, Hindersson P, Steinaa L, Waterston AM, et al. Therapeutic antibodies elicited by immunization against TNF-[alpha] Nat Biotech. 1999;17:666–9. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

