Ultrastructural Changes during the Life Cycle of Mycoplasma salivarium in Oral Biopsies from Patients with Oral Leukoplakia
- PMID: 28983467
- PMCID: PMC5613160
- DOI: 10.3389/fcimb.2017.00403
Ultrastructural Changes during the Life Cycle of Mycoplasma salivarium in Oral Biopsies from Patients with Oral Leukoplakia
Abstract
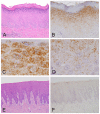
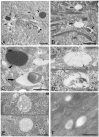
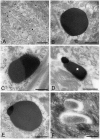
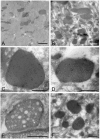
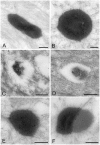
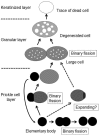
Bacteria in genus Mycoplasma spp. are the smallest and simplest form of freely replicating bacteria, with 16 species known to infect humans. In the mouth, M. salivarium is the most frequently identified species. Mycoplasma spp. are parasites with small genomes. Although most of the Mycoplasma spp. that infect humans remain attached to the host cell surface throughout their life cycle, we have previously reported the presence of Mycoplasma salivarium in the epithelial cells of oral leukoplakia and oral lichen planus. However, the mechanism underlying the pathogenicity of M. salivarium has remained unclear. Further studies are needed to identify the process of infection of human cells and the stages in the life cycle of M. salivarium. Electron microscopy (EM) is the method of choice for morphological investigation of Mycoplasma spp. in cells or tissues. This study was performed to clarify and detail the ultrastructure of M. salivarium in tissue biopsies of oral mucosal leukoplakia, using three EM methods: (1) a standard EM processing method; (2) an ultracryotomy and immunolabeling method; and (3) the LR White resin post-embedding and immunolabeling method. This study included five oral leukoplakia tissue samples showing hyperplasia and hyperkeratosis. Although there was some variation in ultrastructural appearances between the three EM methods used, there were four ultrastructural appearances that are believed to reflect the stages of the M. salivarium life cycle in the epithelial cells of the oral mucosa: (1) small, electron-dense cellular-like structures or elementary bodies of M. salivarium; (2) large structures of M. salivarium; (3) M. salivarium organisms in cell division; (4) the sequence of events in the life cycle of M. salivarium that includes: (a) elementary bodies of M. salivarium deep in the oral mucosal epithelium; (b) replication by binary fission and daughter cell division from the elementary bodies; (c) maturation or degeneration of M. salivarium in the epithelial cells mainly in the upper part of the epithelium; and (d) death of the organisms in the granular and/or keratinized layer. These ultrastructural images may provide a useful reference for the identification of M. salivarium in diagnostic cytology or biopsy material.
Keywords: Mycoplasma; Mycoplasma salivarium; electron microscopy; immunoelectron microscopy; infection; life cycle; oral leukoplakia; oral mucosa.
Figures



Similar articles
-
In situ immunohistochemical detection of intracellular Mycoplasma salivarium in the epithelial cells of oral leukoplakia.J Oral Pathol Med. 2015 Feb;44(2):134-44. doi: 10.1111/jop.12215. Epub 2014 Jul 28. J Oral Pathol Med. 2015. PMID: 25065471 Free PMC article.
-
Immunohistochemical detection of Mycoplasma salivarium in oral lichen planus tissue.J Oral Pathol Med. 2017 Sep;46(8):649-656. doi: 10.1111/jop.12568. Epub 2017 Apr 6. J Oral Pathol Med. 2017. PMID: 28295632 Free PMC article.
-
Mycoplasma salivarium as a dominant coloniser of Fanconi anaemia associated oral carcinoma.PLoS One. 2014 Mar 18;9(3):e92297. doi: 10.1371/journal.pone.0092297. eCollection 2014. PLoS One. 2014. PMID: 24642836 Free PMC article.
-
Leukoplakia: still a gallimaufry or is progress being made?--A review.Adv Anat Pathol. 1998 May;5(3):137-55. Adv Anat Pathol. 1998. PMID: 9868520 Review.
-
A theoretical consideration of some biological parameters involved in cell kinetic investigations of oral leukoplakia and abnormal states in stratified squamous epithelium.J Oral Pathol. 1981 Dec;10(6):375-85. doi: 10.1111/j.1600-0714.1981.tb01289.x. J Oral Pathol. 1981. PMID: 6801233 Review. No abstract available.
Cited by
-
[Clinical implications of the genus Mycoplasma].Rev Esp Quimioter. 2021 Jun;34(3):169-184. doi: 10.37201/req/014.2021. Epub 2021 Mar 18. Rev Esp Quimioter. 2021. PMID: 33735544 Free PMC article. Review. Spanish.
-
Simultaneous presence of Mycoplasma salivarium and Tannerella forsythia in the implant sulcus after lateral augmentation with autogenous root grafts is associated with increased sulcus probing depth.PLoS One. 2022 Jul 8;17(7):e0270962. doi: 10.1371/journal.pone.0270962. eCollection 2022. PLoS One. 2022. PMID: 35802644 Free PMC article.
-
A genomic sequence of the type II-A clustered regularly interspaced short palindromic repeats (CRISPR)/CRISPR-associated system in Mycoplasma salivarium strain ATCC 29803.J Oral Microbiol. 2022 Jan 2;14(1):2008153. doi: 10.1080/20002297.2021.2008153. eCollection 2022. J Oral Microbiol. 2022. PMID: 34992734 Free PMC article.
-
Association between salivary microbiota and renal function in renal transplant patients during the perioperative period.Front Microbiol. 2023 Mar 29;14:1122101. doi: 10.3389/fmicb.2023.1122101. eCollection 2023. Front Microbiol. 2023. PMID: 37065138 Free PMC article.
-
On the Oral Microbiome of Oral Potentially Malignant and Malignant Disorders: Dysbiosis, Loss of Diversity, and Pathogens Enrichment.Int J Mol Sci. 2023 Feb 9;24(4):3466. doi: 10.3390/ijms24043466. Int J Mol Sci. 2023. PMID: 36834903 Free PMC article.
References
-
- Blancherd A., Bebear C. M. (2002). Mycoplasmas of humans, in Molecular Biology and Pathogenicity of Mycoplasmas, eds Razin S., Herrmann R. (New York, NY: Academic/Plenum Publishers; ), 45–71.
-
- De Paul A. L., Mukdsi J. H., Petiti J. P., Gutierrez S., Quintar A. A., Maldonado C. A., et al. (2012). Immunoelectron microscopy: a reliable tool for the analysis of cellular processes, in Applications of Immunocytochemistry, ed Dehghani H. (InTech; ).
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

