PARP‑1 may be involved in hydroquinone‑induced apoptosis by poly ADP‑ribosylation of ZO‑2
- PMID: 28983606
- PMCID: PMC5779892
- DOI: 10.3892/mmr.2017.7643
PARP‑1 may be involved in hydroquinone‑induced apoptosis by poly ADP‑ribosylation of ZO‑2
Abstract
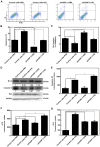
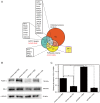
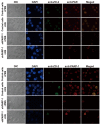
Hydroquinone (HQ), a major reactive metabolite of benzene, contributes to benzene‑induced leukemia. The molecular mechanisms that underlie this activity remain to be elucidated. Poly ADP‑ribosylation (PARylation) is a type of reversible posttranslational modification that is performed by enzymes in the PAR polymerase (PARP) family and mediates different biological processes, including apoptosis. Zona occludens 2 (ZO‑2) is a tight junction scaffold protein, which is involved in cell proliferation and apoptosis. The present study investigated the activity and mechanisms regulated by PARP‑1 during HQ‑induced apoptosis using TK6 lymphoblastoid cells and PARP‑1‑silenced TK6 cells. The results revealed that exposure to 10 µM HQ for 72 h induced apoptosis in TK6 cells and that apoptosis was attenuated in PARP‑1‑silenced TK6 cells. In cells treated with HQ, inhibition of PARP‑1 increased the expression of B cell leukemia/lymphoma 2 (Bcl‑2), increased ATP production and reduced reactive oxygen species (ROS) production relative to the levels observed in cells treated with HQ alone. Co‑localization of ZO‑2 and PAR (or PARP‑1 protein) was determined using immunofluorescence confocal microscopy. The findings of the present study revealed that ZO‑2 was PARylated via an interaction with PARP‑1, which was consistent with an analysis of protein expression that was performed using western blot analysis, which determined that ZO‑2 protein expression was upregulated in HQ‑treated control cells and downregulated in HQ‑treated PARP‑1‑silenced TK6 cells. These findings indicated that prolonged exposure to a low dose of HQ induced TK6 cells to undergo apoptosis, whereas inhibiting PARP‑1 attenuates cellular apoptosis by activating Bcl‑2 and energy‑saving processes and reducing ROS. The present study determined that PARP‑1 was involved in HQ‑induced apoptosis by PARylation of ZO‑2.
Figures

Similar articles
-
Down-regulation of poly (ADP-ribose) polymerase 1 leads to change of hydroquinone cytotoxicity in TK6 cells.Toxicol Mech Methods. 2015;25(6):467-77. doi: 10.3109/15376516.2015.1070222. Epub 2015 Sep 3. Toxicol Mech Methods. 2015. PMID: 26335980
-
Bcl-2 protects TK6 cells against hydroquinone-induced apoptosis through PARP-1 cytoplasm translocation and stabilizing mitochondrial membrane potential.Environ Mol Mutagen. 2018 Jan;59(1):49-59. doi: 10.1002/em.22126. Epub 2017 Aug 26. Environ Mol Mutagen. 2018. PMID: 28843007
-
miR-7-5p overexpression suppresses cell proliferation and promotes apoptosis through inhibiting the ability of DNA damage repair of PARP-1 and BRCA1 in TK6 cells exposed to hydroquinone.Chem Biol Interact. 2018 Mar 1;283:84-90. doi: 10.1016/j.cbi.2018.01.019. Epub 2018 Feb 5. Chem Biol Interact. 2018. PMID: 29421518
-
Hsa_circ_0001944 regulates apoptosis by regulating the binding of PARP1 and HuR in leukemia and malignant transformed cells induced by hydroquinone.Environ Toxicol. 2023 Feb;38(2):381-391. doi: 10.1002/tox.23719. Epub 2022 Nov 30. Environ Toxicol. 2023. PMID: 36448377
-
Inputs and outputs of poly(ADP-ribosyl)ation: Relevance to oxidative stress.Redox Biol. 2014;2:978-82. doi: 10.1016/j.redox.2014.08.003. Epub 2014 Aug 21. Redox Biol. 2014. PMID: 25460733 Free PMC article. Review.
Cited by
-
Protective Effects of Aqueous Extracts of Flos lonicerae Japonicae against Hydroquinone-Induced Toxicity in Hepatic L02 Cells.Oxid Med Cell Longev. 2018 Nov 18;2018:4528581. doi: 10.1155/2018/4528581. eCollection 2018. Oxid Med Cell Longev. 2018. PMID: 30581530 Free PMC article.
-
Striatin is a novel modulator of cell adhesion.FASEB J. 2019 Apr;33(4):4729-4740. doi: 10.1096/fj.201801882R. Epub 2018 Dec 28. FASEB J. 2019. PMID: 30592649 Free PMC article.
-
Brassinin Induces Apoptosis, Autophagy, and Paraptosis via MAPK Signaling Pathway Activation in Chronic Myelogenous Leukemia Cells.Biology (Basel). 2023 Feb 14;12(2):307. doi: 10.3390/biology12020307. Biology (Basel). 2023. PMID: 36829581 Free PMC article.
-
ZO-2 Is a Master Regulator of Gene Expression, Cell Proliferation, Cytoarchitecture, and Cell Size.Int J Mol Sci. 2019 Aug 24;20(17):4128. doi: 10.3390/ijms20174128. Int J Mol Sci. 2019. PMID: 31450555 Free PMC article. Review.
-
Differences in PARP Inhibitors for the Treatment of Ovarian Cancer: Mechanisms of Action, Pharmacology, Safety, and Efficacy.Int J Mol Sci. 2021 Apr 19;22(8):4203. doi: 10.3390/ijms22084203. Int J Mol Sci. 2021. PMID: 33921561 Free PMC article. Review.
References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

