Deficit in Central Auditory Processing as a Biomarker of Pre-Clinical Alzheimer's Disease
- PMID: 28984583
- PMCID: PMC5757649
- DOI: 10.3233/JAD-170545
Deficit in Central Auditory Processing as a Biomarker of Pre-Clinical Alzheimer's Disease
Abstract
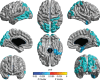
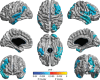
Prevention of dementia due to Alzheimer's disease (d/AD) requires interventions that slow the disease process prior to symptom onset. To develop such interventions, one needs metrics that assess pre-symptomatic disease progression. Familiar measures of progression include cerebrospinal fluid (CSF) biochemical and imaging analyses, as well as cognitive testing. Changes in the latter can sometimes be difficult to distinguish from effects of "normal" aging. A different approach involves testing of "central auditory processing" (CAP), which enables comprehension of auditory stimuli amidst a distracting background (e.g., conversation in a noisy bar or restaurant). Such comprehension is often impaired in d/AD. Similarly, effortful or diminished auditory comprehension is sometimes reported by cognitively healthy elders, raising the possibility that CAP deficit may be a marker of pre-symptomatic AD. In 187 cognitively and physically healthy members of the aging, AD family history-positive PREVENT-AD cohort, we therefore evaluated whether CAP deficits were associated with known markers of AD neurodegeneration. Such markers included CSF tau concentrations and magnetic resonance imaging volumetric and cortical thickness measures in key AD-related regions. Adjusting for age, sex, education, pure-tone hearing, and APOEɛ4 status, we observed a persistent relationship between CAP scores and CSF tau levels, entorhinal and hippocampal cortex volumes, cortical thickness, and deficits in cognition (Repeatable Battery for Assessment of Neuropsychological Status total score, and several of its index scales). These cross-sectional observations suggest that CAP may serve as a novel metric for pre-symptomatic AD pathogenesis. They are therefore being followed up longitudinally with larger samples.
Keywords: Biomarkers; central auditory processing disorder; cognitive function; pre-clinical Alzheimer’s disease; prevention; sensorineural assessment.
Figures
References
-
- Bateman RJ, Xiong C, Benzinger TL, Fagan AM, Goate A, Fox NC, Marcus DS, Cairns NJ, Xie X, Blazey TM, Holtzman DM, Santacruz A, Buckles V, Oliver A, Moulder K, Aisen PS, Ghetti B, Klunk WE, McDade E, Martins RN, Masters CL, Mayeux R, Ringman JM, Rossor MN, Schofield PR, Sperling RA, Salloway S, Morris JC; Dominantly Inherited Alzheimer Network (2012) Clinical and biomarker changes in dominantly inherited Alzheimer’s disease. N Engl J Med 367, 795–804. - PMC - PubMed
-
- Sperling RA, Aisen PS, Beckett LA, Bennett DA, Craft S, Fagan AM, Iwatsubo T, Jack CR Jr, Kaye J, Montine TJ, Park DC, Reiman EM, Rowe CC, Siemers E, Stern Y, Yaffe K, Carrillo MC, Thies B, Morrison-Bogorad M, Wagster MV, Phelps CH (2011) Toward defining the preclinical stages of Alzheimer’s disease: Recommendations from the National Institute on Aging-Alzheimer’s Association workgroups on diagnostic guidelines for Alzheimer’s disease. Alzheimers Dement 7, 280–292. - PMC - PubMed
-
- Small BJ, Fratiglioni L, Viitanen M, Winblad B, Bäckman L (2000) The course of cognitive impairment in preclinical Alzheimerdisease: Three-and 6-year follow-up of a population-based sample. Arch Neurol 57, 839–844. - PubMed
-
- Morris JC, Storandt M, McKeel DW Jr, Rubin EH, Price JL, Grant EA, Berg L (1996) Cerebral amyloid deposition and diffuse plaques in “normal” aging: Evidence for presymptomatic and very mild Alzheimer’s disease. Neurology 46, 707–719. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

