A Complex Lens for a Complex Eye
- PMID: 28992245
- PMCID: PMC5886344
- DOI: 10.1093/icb/icx116
A Complex Lens for a Complex Eye
Abstract
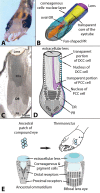
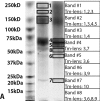
A key innovation for high resolution eyes is a sophisticated lens that precisely focuses light onto photoreceptors. The eyes of holometabolous larvae range from very simple eyes that merely detect light to eyes that are capable of high spatial resolution. Particularly interesting are the bifocal lenses of Thermonectus marmoratus larvae, which differentially focus light on spectrally-distinct retinas. While functional aspects of insect lenses have been relatively well studied, little work has explored their molecular makeup, especially in regard to more complex eye types. To investigate this question, we took a transcriptomic and proteomic approach to identify the major proteins contributing to the principal bifocal lenses of T. marmoratus larvae. Mass spectrometry revealed 10 major lens proteins. Six of these share sequence homology with cuticular proteins, a large class of proteins that are also major components of corneal lenses from adult compound eyes of Drosophila melanogaster and Anopheles gambiae. Two proteins were identified as house-keeping genes and the final two lack any sequence homologies to known genes. Overall the composition seems to follow a pattern of co-opting transparent and optically dense proteins, similar to what has been described for other animal lenses. To identify cells responsible for the secretion of specific lens proteins, we performed in situ hybridization studies and found some expression differences between distal and proximal corneagenous cells. Since the distal cells likely give rise to the periphery and the proximal cells to the center of the lens, our findings highlight a possible mechanism for establishing structural differences that are in line with the bifocal nature of these lenses. A better understanding of lens composition provides insights into the evolution of proper focusing, which is an important step in the transition between low-resolution and high-resolution eyes.
© The Author 2017. Published by Oxford University Press on behalf of the Society for Integrative and Comparative Biology. All rights reserved. For permissions please email: journals.permissions@oup.com.
Figures



References
-
- Altschul SF, Gish W, Miller W, Myers EW, Lipman DJ.. 1990. Basic local alignment search tool. J Mol Biol 215:403–10. - PubMed
-
- Andersen SO, Rafn K, Roepstorff P.. 1997. Sequence studies of proteins from larval and pupal cuticle of the yellow meal worm, Tenebrio molitor. Insect Biochem Mol Biol 27:121–31. - PubMed
-
- Blanco J, Girard F, Kamachi Y, Kondoh H, Gehring WJ.. 2005. Functional analysis of the chicken delta 1-crystallin enhancer activity in Drosophila reveals remarkable evolutionary conservation between chicken and fly. Development 132:1895–905. - PubMed
-
- Bland K, Revetta N, Stowasser A, Buschbeck EK. 2014. Unilateral range finding in diving beetle larvae. J Exp Biol 217:327–30. - PubMed
-
- Blest AD, Land MF.. 1977. Physiological optics of Dinopis subrufus L. Koch: a fish-lens in a spider. Proc R Soc Lond B Biol Sci 196:197–222. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

