Plasma membrane-associated cation-binding protein 1-like protein negatively regulates intercellular movement of BaMV
- PMID: 28992255
- PMCID: PMC5853580
- DOI: 10.1093/jxb/erx307
Plasma membrane-associated cation-binding protein 1-like protein negatively regulates intercellular movement of BaMV
Abstract
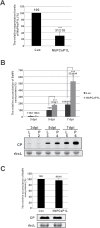
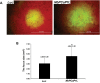
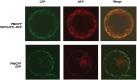
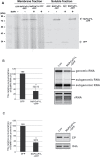
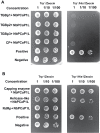
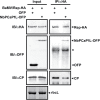
To establish a successful infection, a virus needs to replicate and move cell-to-cell efficiently. We investigated whether one of the genes upregulated in Nicotiana benthamiana after Bamboo mosaic virus (BaMV) inoculation was involved in regulating virus movement. We revealed the gene to be a plasma membrane-associated cation-binding protein 1-like protein, designated NbPCaP1L. The expression of NbPCaP1L in N. benthamiana was knocked down using Tobacco rattle virus-based gene silencing and consequently the accumulation of BaMV increased significantly to that of control plants. Further analysis indicated no significant difference in the accumulation of BaMV in NbPCaP1L knockdown and control protoplasts, suggesting NbPCaP1L may affect cell-to-cell movement of BaMV. Using a viral vector expressing green fluorescent protein in the knockdown plants, the mean area of viral focus, as determined by fluorescence, was found to be larger in NbPCaP1L knockdown plants. Orange fluorescence protein (OFP)-fused NbPCaP1L, NbPCaP1L-OFP, was expressed in N. benthamiana and reduced the accumulation of BaMV to 46%. To reveal the possible interaction of viral protein with NbPCaP1L, we performed yeast two-hybrid and co-immunoprecipitation experiments. The results indicated that NbPCaP1L interacted with BaMV replicase. The results also suggested that NbPCaP1L could trap the BaMV movement RNP complex via interaction with the viral replicase in the complex and so restricted viral cell-to-cell movement.
Keywords: Bamboo mosaic virus; NbPCaP1L; Nicotiana benthamiana; defense protein; positive-sense RNA virus; replicase; viral RNA movement.
© The Author 2017. Published by Oxford University Press on behalf of the Society for Experimental Biology.
Figures
References
-
- Agre P, Bonhivers M, Borgnia MJ. 1998. The aquaporins, blueprints for cellular plumbing systems. The Journal of Biological Chemistry 273, 14659–14662. - PubMed
-
- Baurès I, Candresse T, Leveau A, Bendahmane A, Sturbois B. 2008. The Rx gene confers resistance to a range of potexviruses in transgenic Nicotiana plants. Molecular Plant-Microbe Interactions 21, 1154–1164. - PubMed
-
- Benitez-Alfonso Y, Faulkner C, Ritzenthaler C, Maule AJ. 2010. Plasmodesmata: gateways to local and systemic virus infection. Molecular Plant-Microbe Interactions 23, 1403–1412. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

