Evaluation of a Plasmodium-Specific Carrier Protein To Enhance Production of Recombinant Pf s25, a Leading Transmission-Blocking Vaccine Candidate
- PMID: 28993460
- PMCID: PMC5736822
- DOI: 10.1128/IAI.00486-17
Evaluation of a Plasmodium-Specific Carrier Protein To Enhance Production of Recombinant Pf s25, a Leading Transmission-Blocking Vaccine Candidate
Abstract
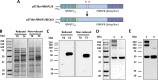
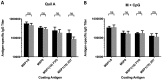
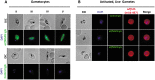
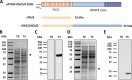
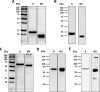
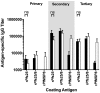
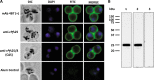
Challenges with the production and suboptimal immunogenicity of malaria vaccine candidates have slowed the development of a Plasmodium falciparum multiantigen vaccine. Attempting to resolve these issues, we focused on the use of highly immunogenic merozoite surface protein 8 (MSP8) as a vaccine carrier protein. Previously, we showed that a genetic fusion of the C-terminal 19-kDa fragment of merozoite surface protein 1 (MSP119) to P. falciparum MSP8 (PfMSP8) facilitated antigen production and folding and the induction of neutralizing antibodies to conformational B cell epitopes of MSP119 Here, using the PfMSP1/8 construct, we further optimized the recombinant PfMSP8 (rPfMSP8) carrier by the introduction of two cysteine-to-serine substitutions (CΔS) to improve the yield of the monomeric product. We then sought to test the broad applicability of this approach using the transmission-blocking vaccine candidate Pfs25. The production of rPfs25-based vaccines has presented challenges. Antibodies directed against the four highly constrained epidermal growth factor (EGF)-like domains of Pfs25 block sexual-stage development in mosquitoes. The sequence encoding mature Pfs25 was codon harmonized for expression in Escherichia coli We produced a rPfs25-PfMSP8 fusion protein [rPfs25/8(CΔS)] as well as unfused, mature rPfs25. rPfs25 was purified with a modest yield but required the incorporation of refolding protocols to obtain a proper conformation. In comparison, chimeric rPfs25/8(CΔS) was expressed and easily purified, with the Pfs25 domain bearing the proper conformation without renaturation. Both antigens were immunogenic in rabbits, inducing IgG that bound native Pfs25 and exhibited potent transmission-reducing activity. These data further demonstrate the utility of PfMSP8 as a parasite-specific carrier protein to enhance the production of complex malaria vaccine targets.
Keywords: MSP8 carrier protein; malaria; subunit vaccines; transmission blocking.
Copyright © 2017 American Society for Microbiology.
Figures

Similar articles
-
Maintaining immunogenicity of blood stage and sexual stage subunit malaria vaccines when formulated in combination.PLoS One. 2020 Apr 29;15(4):e0232355. doi: 10.1371/journal.pone.0232355. eCollection 2020. PLoS One. 2020. PMID: 32348377 Free PMC article.
-
Immunogenicity of a chimeric Plasmodium falciparum merozoite surface protein vaccine in Aotus monkeys.Malar J. 2016 Mar 15;15:159. doi: 10.1186/s12936-016-1226-5. Malar J. 2016. PMID: 26975721 Free PMC article.
-
Evaluation of the immunogenicity and vaccine potential of recombinant Plasmodium falciparum merozoite surface protein 8.Infect Immun. 2012 Jul;80(7):2473-84. doi: 10.1128/IAI.00211-12. Epub 2012 May 14. Infect Immun. 2012. PMID: 22585960 Free PMC article.
-
Immunogenicity of Plasmodium falciparum sexual stage antigens: implications for the design of a transmission blocking vaccine.Immunol Lett. 1990 Aug;25(1-3):83-6. doi: 10.1016/0165-2478(90)90096-9. Immunol Lett. 1990. PMID: 1704352 Review.
-
Strategies & recent development of transmission-blocking vaccines against Plasmodium falciparum.Indian J Med Res. 2016 Jun;143(6):696-711. doi: 10.4103/0971-5916.191927. Indian J Med Res. 2016. PMID: 27748294 Free PMC article. Review.
Cited by
-
Inclusion of an Optimized Plasmodium falciparum Merozoite Surface Protein 2-Based Antigen in a Trivalent, Multistage Malaria Vaccine.J Immunol. 2021 Apr 15;206(8):1817-1831. doi: 10.4049/jimmunol.2000927. Epub 2021 Mar 31. J Immunol. 2021. PMID: 33789984 Free PMC article.
-
Malaria transmission-blocking vaccines: wheat germ cell-free technology can accelerate vaccine development.Expert Rev Vaccines. 2019 Oct;18(10):1017-1027. doi: 10.1080/14760584.2019.1674145. Epub 2019 Oct 10. Expert Rev Vaccines. 2019. PMID: 31566026 Free PMC article. Review.
-
In vivo Characterization of Plasmodium berghei P47 (Pbs47) as a Malaria Transmission-Blocking Vaccine Target.Front Microbiol. 2020 Jul 3;11:1496. doi: 10.3389/fmicb.2020.01496. eCollection 2020. Front Microbiol. 2020. PMID: 32719666 Free PMC article.
-
A Multi-Stage Plasmodium vivax Malaria Vaccine Candidate Able to Induce Long-Lived Antibody Responses Against Blood Stage Parasites and Robust Transmission-Blocking Activity.Front Cell Infect Microbiol. 2019 May 1;9:135. doi: 10.3389/fcimb.2019.00135. eCollection 2019. Front Cell Infect Microbiol. 2019. PMID: 31119106 Free PMC article.
-
Transmission-Blocking Vaccines: Old Friends and New Prospects.Infect Immun. 2019 May 21;87(6):e00775-18. doi: 10.1128/IAI.00775-18. Print 2019 Jun. Infect Immun. 2019. PMID: 30962400 Free PMC article. Review.
References
-
- Anonymous. 2016. World malaria report 2016. World Health Organization, Geneva, Switzerland.
-
- RTS,S Clinical Trials Partnership, Agnandji ST, Lell B, Fernandes JF, Abossolo BP, Methogo BG, Kabwende AL, Adegnika AA, Mordmuller B, Issifou S, Kremsner PG, Sacarlal J, Aide P, Lanaspa M, Aponte JJ, Machevo S, Acacio S, Bulo H, Sigauque B, Macete E, Alonso P, Abdulla S, Salim N, Minja R, Mpina M, Ahmed S, Ali AM, Mtoro AT, Hamad AS, Mutani P, Tanner M, Tinto H, D'Alessandro U, Sorgho H, Valea I, Bihoun B, Guiraud I, Kabore B, Sombie O, Guiguemde RT, Ouedraogo JB, Hamel MJ, Kariuki S, Oneko M, Odero C, Otieno K, Awino N, McMorrow M, Muturi-Kioi V, Laserson KF, et al. . 2012. A phase 3 trial of RTS,S/AS01 malaria vaccine in African infants. N Engl J Med 367:2284–2295. doi:10.1056/NEJMoa1208394. - DOI - PMC - PubMed
-
- RTS,S Clinical Trials Partnership. 2015. Efficacy and safety of RTS,S/AS01 malaria vaccine with or without a booster dose in infants and children in Africa: final results of a phase 3, individually randomised, controlled trial. Lancet 386:31–45. doi:10.1016/S0140-6736(15)60721-8. - DOI - PMC - PubMed
-
- White MT, Verity R, Griffin JT, Asante KP, Owusu-Agyei S, Greenwood B, Drakeley C, Gesase S, Lusingu J, Ansong D, Adjei S, Agbenyega T, Ogutu B, Otieno L, Otieno W, Agnandji ST, Lell B, Kremsner P, Hoffman I, Martinson F, Kamthunzu P, Tinto H, Valea I, Sorgho H, Oneko M, Otieno K, Hamel MJ, Salim N, Mtoro A, Abdulla S, Aide P, Sacarlal J, Aponte JJ, Njuguna P, Marsh K, Bejon P, Riley EM, Ghani AC. 2015. Immunogenicity of the RTS,S/AS01 malaria vaccine and implications for duration of vaccine efficacy: secondary analysis of data from a phase 3 randomised controlled trial. Lancet Infect Dis 15:1450–1458. doi:10.1016/S1473-3099(15)00239-X. - DOI - PMC - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

