Contribution of dietary intake to relapse rate in early paediatric multiple sclerosis
- PMID: 28993476
- PMCID: PMC5732893
- DOI: 10.1136/jnnp-2017-315936
Contribution of dietary intake to relapse rate in early paediatric multiple sclerosis
Abstract
Objective: The role of diet in multiple sclerosis (MS) course remains largely unknown. Children with MS have a higher relapse rate compared with MS in adults. Thus, studying the effect of diet on relapse rate in this age group is likely to provide more robust answers.
Methods: This is a multicentre study done at 11 paediatric MS centres in the USA. Patients with relapsing-remitting MS (RRMS) or clinically isolated syndrome (CIS) with disease onset before 18 years of age and duration of less than 4 years were included in this study. Dietary intake during the week before enrolment was assessed with the validated Block Kids Food Screener. The outcome of the study was time from enrolment to the next relapse. 219 patients with paediatric RRMS or CIS were enrolled. Each 10% increase in energy intake from fat increased the hazard of relapse by 56% (adjusted HR 1.56, 95% CI 1.05 to 2.31, p=0.027), and in particular each 10% increase in saturated fat tripled this hazard (adjusted HR: 3.37, 95% CI 1.34 to 8.43, p=0.009). In contrast, each additional one cup equivalent of vegetable decreased the hazard of relapse by 50% (adjusted HR: 0.50, 95% CI 0.27 to 0.91, p=0.024). These associations remained with mutual adjustment and persisted when adjusting for baseline 25(OH) vitamin D serum level. Other studied nutrients were not associated with relapse.
Conclusions: This study suggests that in children with MS, high energy intake from fat, especially saturated fat, may increase the hazard to relapse, while vegetable intake may be independently protective.
Keywords: Diet; Fat intake; Multiple sclerosis; Pediatric; Vegetable intake; relapse.
© Article author(s) (or their employer(s) unless otherwise stated in the text of the article) 2018. All rights reserved. No commercial use is permitted unless otherwise expressly granted.
Conflict of interest statement
Competing interests: Dr JG was supported by grants from Race to Erase MS, NMSS, Biogen and Genentech during this work. Dr AW is funded by the NIH (NINDS, K23NS069806) and has received research funding from Biogen Idec. Dr CTC has been supported by the National MS Society and the NIH (R01NS071463). He is an ad hoc consultant for Biovest International, Inc. Dr EW is funded by the National MS Society, the NIH and the Race to Erase MS. She volunteers on an advisory board for a clinical trial of Novartis. Dr LK is supported by the National MS Society, NIH, Robert and Lisa Lourie Foundation, Department of Defense. She has received honoraria, consulting payments, grant support or royalties from Biogen, Medimmune, Novartis, Teva Neuroscience, Sanofi-Aventis and EMD Serono. Dr BWG received honoraria for serving in advisory boards and educational programmes from Teva Pharmaceuticals, Biogen Idec, Novartis, Acorda EMD Serono, Novartis, Genzyme and Sanofi. She also received support for research activities from the National Institutes of Health, National Multiple Sclerosis Society, National Science Foundation, Department of Defense, EMD Serono, Biogen Idec, Teva Neuroscience, Novartis, Acorda, Genzyme and the Jog for the Jake Foundation. Dr TC has served as a consultant for Biogen Idec, Teva Neurosciences, Novartis and Sanofi-Aventis and has received grant support from NIH, National MS Society, Guthy-Jackson Charitable Foundation, CMSC and Merck-Serono, Novartis, and Biogen and Verily. Dr JR has research funding from Teva Neuroscience and Biogen. He is a member of the Medical Advisory Board for the DECIDE trial, which is funded by Biogen and AbbVie. Dr GA has received research support from Biogen-Idec. Drs AB, JN, MG, TL, MR and GA have no disclosures. JH has no disclosures. Dr J-MT received funding from the NIH (NCATS) during this work.
Figures
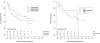
Comment in
-
Diet and disease modification in multiple sclerosis: a nutritional epidemiology perspective.J Neurol Neurosurg Psychiatry. 2018 Jan;89(1):3. doi: 10.1136/jnnp-2017-316375. Epub 2017 Oct 9. J Neurol Neurosurg Psychiatry. 2018. PMID: 28993475 No abstract available.
References
-
- NutritionQuest. [accessed Oct 1 2007];Assessment Tools. Block ’98 FFQ. 2017 http://www.nutritionquest.com/products/questionnaires_screeners.htm.
-
- Ascherio A. Environmental factors in multiple sclerosis. Expert Rev Neurother. 2013;13:3–9. - PubMed
-
- Weston M, Constantinescu CS. What role does tobacco smoking play in multiple sclerosis disability and mortality? A review of the evidence. Neurodegener Dis Manag. 2015;5:19–25. - PubMed
-
- Mowry EM, Krupp LB, Milazzo M, et al. Vitamin D status is associated with relapse rate in pediatric-onset multiple sclerosis. Ann Neurol. 2010;67:618–24. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
