Carboxyethylgermanium sesquioxide (Ge-132) treatment during in vitro culture protects fertilized porcine embryos against oxidative stress induced apoptosis
- PMID: 28993559
- PMCID: PMC5735269
- DOI: 10.1262/jrd.2017-020
Carboxyethylgermanium sesquioxide (Ge-132) treatment during in vitro culture protects fertilized porcine embryos against oxidative stress induced apoptosis
Abstract
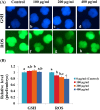
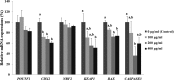
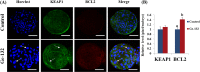
Compared with the in vivo environment, porcine in vitro embryo-culture systems are suboptimal, as they induce oxidative stress via the accumulation of reactive oxygen species (ROS). High ROS levels during early embryonic development cause negative effects, such as apoptosis. In this study, we examined the effects of the antioxidant carboxyethylgermanium sesquioxide (Ge-132) during in vitro culture (IVC) on embryonic development in porcine in vitro fertilization (IVF) embryos. Zygotes were treated with different concentrations of Ge-132 (0, 100, 200 and 400 μg/ml). All of the Ge-132 treatment groups displayed greater total cell numbers after IVC (98.1, 98.5 and 103.4, respectively) compared with the control group (73.9). The 200 μg/ml Ge-132 treatment group exhibited significantly increased intracellular GSH levels compared with the control group, whereas the ROS generation levels decreased in Ge-132 dose-dependent manner (P < 0.05). The mRNA expression levels of the KEAP1 gene and proapoptotic genes BAX and CASPASE3 were lower in the Ge-132 treated blastocysts compared with the control group (P < 0.05). The percentages of apoptotic and necrotic cells in the Ge-132 treated embryos on day 2 (48 h) were significantly lower than the untreated embryos (9.1 vs. 17.1% and 0 vs. 2.7%, respectively). In the day 7 blastocysts, the percentages of apoptotic cells in 200 µg/ml Ge-132 treated group were lower compared to controls (1.6 vs. 2.5%). More KEAP1 protein was found to be localized in cytoplasm of the 200 μg/ml Ge-132 treated blastocysts, whereas KEAP1 protein was predominantly nuclei in the control blastocysts. These results indicate that the developmental competence of embryos cultured under Ge-132 treatment may be associated with KEAP1 signaling cascades involved in oxidative stress and apoptosis during porcine preimplantation embryo development.
Keywords: Apoptosis; Ge-132; In vitro culture (IVC) porcine embryos; Oxidative stress.
Figures
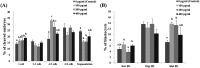
Similar articles
-
Antioxidative effect of carboxyethylgermanium sesquioxide (Ge-132) on IVM of porcine oocytes and subsequent embryonic development after parthenogenetic activation and IVF.Theriogenology. 2015 Jul 15;84(2):226-36. doi: 10.1016/j.theriogenology.2015.03.006. Epub 2015 Mar 19. Theriogenology. 2015. PMID: 25913277
-
Exogenous γ-tocotrienol promotes preimplantation development and improves the quality of porcine embryos.Reprod Fertil Dev. 2015 Mar;27(3):481-90. doi: 10.1071/RD13167. Reprod Fertil Dev. 2015. PMID: 24411581
-
Development to the blastocyst stage, the oxidative state, and the quality of early developmental stage of porcine embryos cultured in alteration of glucose concentrations in vitro under different oxygen tensions.Reprod Biol Endocrinol. 2006 Nov 6;4:54. doi: 10.1186/1477-7827-4-54. Reprod Biol Endocrinol. 2006. PMID: 17087833 Free PMC article.
-
Embryotropic effects of vascular endothelial growth factor on porcine embryos produced by in vitro fertilization.Theriogenology. 2018 Oct 15;120:147-156. doi: 10.1016/j.theriogenology.2018.07.024. Epub 2018 Aug 16. Theriogenology. 2018. PMID: 30121547
-
Mitochondria-targeted therapy rescues development and quality of embryos derived from oocytes matured under oxidative stress conditions: a bovine in vitro model.Hum Reprod. 2019 Oct 2;34(10):1984-1998. doi: 10.1093/humrep/dez161. Hum Reprod. 2019. PMID: 31625574
Cited by
-
Trace elements exposure affects the outcomes of in vitro fertilization embryo transfer, a cohort study in Northern China.J Assist Reprod Genet. 2024 Dec;41(12):3405-3414. doi: 10.1007/s10815-024-03300-9. Epub 2024 Oct 30. J Assist Reprod Genet. 2024. PMID: 39477908
-
The Organogermanium Compound THGP Suppresses Melanin Synthesis via Complex Formation with L-DOPA on Mushroom Tyrosinase and in B16 4A5 Melanoma Cells.Int J Mol Sci. 2019 Sep 26;20(19):4785. doi: 10.3390/ijms20194785. Int J Mol Sci. 2019. PMID: 31561511 Free PMC article.
-
Physiological Activity of Trace Element Germanium including Anticancer Properties.Biomedicines. 2023 May 25;11(6):1535. doi: 10.3390/biomedicines11061535. Biomedicines. 2023. PMID: 37371629 Free PMC article. Review.
-
The role of germanium in diseases: exploring its important biological effects.J Transl Med. 2023 Nov 8;21(1):795. doi: 10.1186/s12967-023-04643-0. J Transl Med. 2023. PMID: 37940963 Free PMC article.
-
Exogenous Melatonin in the Culture Medium Does Not Affect the Development of In Vivo-Derived Pig Embryos but Substantially Improves the Quality of In Vitro-Produced Embryos.Antioxidants (Basel). 2022 Jun 15;11(6):1177. doi: 10.3390/antiox11061177. Antioxidants (Basel). 2022. PMID: 35740074 Free PMC article.
References
-
- Macháty Z, Day BN, Prather RS. Development of early porcine embryos in vitro and in vivo. Biol Reprod 1998; 59: 451–455. - PubMed
-
- Agarwal A, Allamaneni SS, Nallella KP, George AT, Mascha E. Correlation of reactive oxygen species levels with the fertilization rate after in vitro fertilization: a qualified meta-analysis. Fertil Steril 2005; 84: 228–231. - PubMed
-
- Gadea J. Sperm factors related to in vitro and in vivo porcine fertility. Theriogenology 2005; 63: 431–444. - PubMed
-
- Guérin P, El Mouatassim S, Ménézo Y. Oxidative stress and protection against reactive oxygen species in the pre-implantation embryo and its surroundings. Hum Reprod Update 2001; 7: 175–189. - PubMed
-
- Dennery PA. Effects of oxidative stress on embryonic development. Birth Defects Res C Embryo Today 2007; 81: 155–162. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

