Methotrexate versus cyclophosphamide for remission maintenance in ANCA-associated vasculitis: A randomised trial
- PMID: 29016646
- PMCID: PMC5634660
- DOI: 10.1371/journal.pone.0185880
Methotrexate versus cyclophosphamide for remission maintenance in ANCA-associated vasculitis: A randomised trial
Abstract
Objectives: The treatment of anti-neutrophil cytoplasmic antibody (ANCA)-associated vasculitis (AAV) is based on remission-induction and remission-maintenance. Methotrexate is a widely used immunosuppressant but only a few studies explored its role for maintenance in AAV. This trial investigated the efficacy and safety of methotrexate as maintenance therapy for AAV.
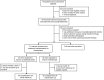
Methods: In this single-centre, open-label, randomised trial we compared methotrexate and cyclophosphamide for maintenance in AAV. We enrolled patients with granulomatosis with polyangiitis (GPA), microscopic polyangiitis (MPA) and eosinophilic granulomatosis with polyangiitis (EGPA), the latter with poor-prognosis factors and/or peripheral neuropathy. Remission was induced with cyclophosphamide. At remission, the patients were randomised to receive methotrexate or to continue with cyclophosphamide for 12 months; after treatment, they were followed for another 12 months. The primary end-point was relapse; secondary end-points included renal outcomes and treatment-related toxicity.
Results: Of the 94 enrolled patients, 23 were excluded during remission-induction or did not achieve remission; the remaining 71 were randomised to cyclophosphamide (n = 33) or methotrexate (n = 38). Relapse frequencies at months 12 and 24 after randomisation were not different between the two groups (p = 1.00 and 1.00). Relapse-free survival was also comparable (log-rank test p = 0.99). No differences in relapses were detected between the two treatments when GPA+MPA and EGPA were analysed separately. There were no differences in eGFR at months 12 and 24; proteinuria declined significantly (from diagnosis to month 24) only in the cyclophosphamide group (p = 0.0007). No significant differences in adverse event frequencies were observed.
Conclusions: MTX may be effective and safe for remission-maintenance in AAV.
Trial registration: clinicaltrials.gov NCT00751517.
Conflict of interest statement
Figures


References
-
- Jennette JC, Falk RJ, Andrassy K, Bacon PA, Churg J, Gross WL, et al. Nomenclature of systemic vasculitides. Proposal of an international consensus conference. Arthritis and rheumatism. 1994;37(2):187–92. Epub 1994/02/01. . - PubMed
-
- Mahr A, Katsahian S, Varet H, Guillevin L, Hagen EC, Hoglund P, et al. Revisiting the classification of clinical phenotypes of anti-neutrophil cytoplasmic antibody-associated vasculitis: a cluster analysis. Annals of the rheumatic diseases. 2013;72(6):1003–10. Epub 2012/09/11. doi: 10.1136/annrheumdis-2012-201750 . - DOI - PubMed
-
- Sinico RA, Di Toma L, Maggiore U, Bottero P, Radice A, Tosoni C, et al. Prevalence and clinical significance of antineutrophil cytoplasmic antibodies in Churg-Strauss syndrome. Arthritis and rheumatism. 2005;52(9):2926–35. Epub 2005/09/06. doi: 10.1002/art.21250 . - DOI - PubMed
-
- Jayne D. Update on the European Vasculitis Study Group trials. Current opinion in rheumatology. 2001;13(1):48–55. Epub 2001/01/10. . - PubMed
-
- Cohen P, Pagnoux C, Mahr A, Arene JP, Mouthon L, Le Guern V, et al. Churg-Strauss syndrome with poor-prognosis factors: A prospective multicenter trial comparing glucocorticoids and six or twelve cyclophosphamide pulses in forty-eight patients. Arthritis and rheumatism. 2007;57(4):686–93. Epub 2007/05/02. doi: 10.1002/art.22679 . - DOI - PubMed
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

