Multicenter pilot study of radiochemotherapy as first-line treatment for adults with medulloblastoma (NOA-07)
- PMID: 29016837
- PMCID: PMC5817955
- DOI: 10.1093/neuonc/nox155
Multicenter pilot study of radiochemotherapy as first-line treatment for adults with medulloblastoma (NOA-07)
Erratum in
-
Corrigendum.Neuro Oncol. 2018 Feb 19;20(3):434. doi: 10.1093/neuonc/noy005. Neuro Oncol. 2018. PMID: 29471447 Free PMC article. No abstract available.
Abstract
Background: Medulloblastoma in adult patients is rare, with 0.6 cases per million. Prognosis depends on clinical factors and medulloblastoma entity. No prospective data on the feasibility of radiochemotherapy exist. The German Neuro-Oncology Working Group (NOA) performed a prospective descriptive multicenter single-arm phase II trial to evaluate feasibility and toxicity of radio-polychemotherapy.
Methods: The NOA-07 trial combined craniospinal irradiation with vincristine, followed by 8 cycles of cisplatin, lomustine, and vincristine. Adverse events, imaging and progression patterns, histological and genetic markers, health-related quality of life (HRQoL), and cognition were evaluated. Primary endpoint was the rate of toxicity-related treatment terminations after 4 chemotherapy cycles, and the toxicity profile. The feasibility goal was reached if at least 45% of patients received at least 4 cycles of maintenance chemotherapy.
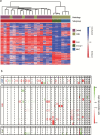
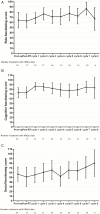
Results: Thirty patients were evaluable. Each 50% showed classic and desmoplastic/nodular histology. Sixty-seven percent were classified into the sonic hedgehog (SHH) subgroup without TP53 alterations, 13% in wingless (WNT), and 17% in non-WNT/non-SHH. Four cycles of chemotherapy were feasible in the majority (n = 21; 70.0%). Hematological side effects and polyneuropathy were prevalent toxicities. During the active treatment period, HRQoL and verbal fluency improved significantly. The 3-year event-free survival rate was 66.6% at the time of databank lock.
Conclusions: Radio-polychemotherapy did lead to considerable toxicity and a high amount of dose reductions throughout the first 4 chemotherapy cycles that may affect efficacy. Thus, we propose frequent patient surveillance using this regimen. Modifications of the regimen may increase feasibility of radio-polychemotherapy of adult patients with medulloblastoma.
© The Author(s) 2017. Published by Oxford University Press on behalf of the Society for Neuro-Oncology. All rights reserved. For permissions, please e-mail: journals.permissions@oup.com
Figures
References
-
- Peris-Bonet R, Martinez-Garcia C, Lacour B et al. Childhood central nervous system tumours—incidence and survival in Europe (1978–1997): report from the Automated Childhood Cancer Information System project. Eur J Cancer. 2006;42(13):2064–2080. - PubMed
-
- Smoll NR. Relative survival of childhood and adult medulloblastomas and primitive neuroectodermal tumors (PNETs). Cancer. 2012;118(5):1313–1322. - PubMed
-
- Chang CH, Housepian EM, Herbert C Jr. An operative staging system and a megavoltage radiotherapeutic technic for cerebellar medulloblastomas. Radiology. 1969;93(6):1351–1359. - PubMed
-
- Remke M, Hielscher T, Northcott PA et al. Adult medulloblastoma comprises three major molecular variants. J Clin Oncol. 2011;29(19):2717–2723. - PubMed
-
- Tabori U, Sung L, Hukin J et al. ; Canadian Pediatric Brain Tumor Consortium Medulloblastoma in the second decade of life: a specific group with respect to toxicity and management: a Canadian Pediatric Brain Tumor Consortium Study. Cancer. 2005;103(9):1874–1880. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

