Mechanistic Evaluation and Translational Signature of Gemcitabine-induced Chemoresistance by Quantitative Phosphoproteomics Analysis with iTRAQ Labeling Mass Spectrometry
- PMID: 29018223
- PMCID: PMC5634998
- DOI: 10.1038/s41598-017-13330-2
Mechanistic Evaluation and Translational Signature of Gemcitabine-induced Chemoresistance by Quantitative Phosphoproteomics Analysis with iTRAQ Labeling Mass Spectrometry
Abstract
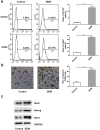
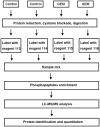
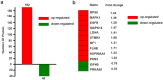
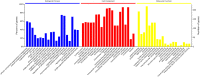
One of the main causations of the poor prognosis of pancreatic cancer is the lack of effective chemotherapies. Gemcitabine is a widely used chemotherapeutic drug, but limited therapeutic efficacy is achieved due to chemoresistance. Recent studies demonstrated that the presence of cancer stem cells may lead to the failure of chemotherapy. Moreover, gemcitabine can promote the stemness of pancreatic cancer cells. We detected the alterations in protein phosphorylation and signaling pathways in pancreatic cancer cells after gemcitabine treatment using iTRAQ labeling LC-MS/MS, because it was featured with the advantages of strong separation ability and analysis range. A total of 232 differentially expressed phosphorylated proteins were identified in this study. Gene Ontology analysis revealed that nuclear lumen, nuclear part and organelle lumen were enriched for cell components and protein binding, poly (A) RNA binding and RNA binding were enriched for molecular function. A variety of signaling pathways were enriched based on KEGG analysis. AMPK, mTOR and PI3K/Akt pathways were verified after gemcitabine exposure. Moreover, we found there were complex interactions of phosphorylated proteins in modulating cancer stemness induced by gemcitabine exposure based on PPIs map. Our experiments may identify potential targets and strategies for sensitizing pancreatic cancer cells to gemcitabine.
Conflict of interest statement
The authors declare that they have no competing interests.
Figures


Similar articles
-
Hypoxia potentiates gemcitabine-induced stemness in pancreatic cancer cells through AKT/Notch1 signaling.J Exp Clin Cancer Res. 2018 Nov 28;37(1):291. doi: 10.1186/s13046-018-0972-3. J Exp Clin Cancer Res. 2018. PMID: 30486896 Free PMC article.
-
Gemcitabine treatment promotes pancreatic cancer stemness through the Nox/ROS/NF-κB/STAT3 signaling cascade.Cancer Lett. 2016 Nov 1;382(1):53-63. doi: 10.1016/j.canlet.2016.08.023. Epub 2016 Aug 26. Cancer Lett. 2016. PMID: 27576197
-
Thymoquinone Pretreatment Overcomes the Insensitivity and Potentiates the Antitumor Effect of Gemcitabine Through Abrogation of Notch1, PI3K/Akt/mTOR Regulated Signaling Pathways in Pancreatic Cancer.Dig Dis Sci. 2015 Apr;60(4):1067-80. doi: 10.1007/s10620-014-3394-x. Epub 2014 Oct 26. Dig Dis Sci. 2015. PMID: 25344906
-
Gemcitabine: metabolism and molecular mechanisms of action, sensitivity and chemoresistance in pancreatic cancer.Eur J Pharmacol. 2014 Oct 15;741:8-16. doi: 10.1016/j.ejphar.2014.07.041. Epub 2014 Jul 30. Eur J Pharmacol. 2014. PMID: 25084222 Review.
-
Gemcitabine resistance in pancreatic ductal adenocarcinoma.Drug Resist Updat. 2015 Nov;23:55-68. doi: 10.1016/j.drup.2015.10.002. Epub 2015 Nov 3. Drug Resist Updat. 2015. PMID: 26690340 Review.
Cited by
-
ZIP4 Increases Expression of Transcription Factor ZEB1 to Promote Integrin α3β1 Signaling and Inhibit Expression of the Gemcitabine Transporter ENT1 in Pancreatic Cancer Cells.Gastroenterology. 2020 Feb;158(3):679-692.e1. doi: 10.1053/j.gastro.2019.10.038. Epub 2019 Nov 9. Gastroenterology. 2020. PMID: 31711924 Free PMC article.
-
Effect of Praziquantel on Schistosoma mekongi Proteome and Phosphoproteome.Pathogens. 2020 May 27;9(6):417. doi: 10.3390/pathogens9060417. Pathogens. 2020. PMID: 32471184 Free PMC article.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

