A chemical and biological toolbox for Type Vd secretion: Characterization of the phospholipase A1 autotransporter FplA from Fusobacterium nucleatum
- PMID: 29021252
- PMCID: PMC5724010
- DOI: 10.1074/jbc.M117.819144
A chemical and biological toolbox for Type Vd secretion: Characterization of the phospholipase A1 autotransporter FplA from Fusobacterium nucleatum
Abstract
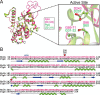
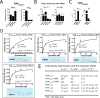
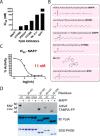
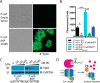
Fusobacterium nucleatum is an oral pathogen that is linked to multiple human infections and colorectal cancer. Strikingly, F. nucleatum achieves virulence in the absence of large, multiprotein secretion systems (Types I, II, III, IV, and VI), which are widely used by Gram-negative bacteria for pathogenesis. By contrast, F. nucleatum strains contain genomic expansions of Type V secreted effectors (autotransporters) that are critical for host cell adherence, invasion, and biofilm formation. Here, we present the first characterization of an F. nucleatum Type Vd phospholipase class A1 autotransporter (strain ATCC 25586, gene FN1704) that we hereby rename Fusobacterium phospholipase autotransporter (FplA). Biochemical analysis of multiple Fusobacterium strains revealed that FplA is expressed as a full-length 85-kDa outer membrane-embedded protein or as a truncated phospholipase domain that remains associated with the outer membrane. Whereas the role of Type Vd secretion in bacteria remains unidentified, we show that FplA binds with high affinity to host phosphoinositide-signaling lipids, revealing a potential role for this enzyme in establishing an F. nucleatum intracellular niche. To further analyze the role of FplA, we developed an fplA gene knock-out strain, which will guide future in vivo studies to determine its potential role in F. nucleatum pathogenesis. In summary, using recombinant FplA constructs, we have identified a biochemical toolbox that includes lipid substrates for enzymatic assays, potent inhibitors, and chemical probes to detect, track, and characterize the role of Type Vd secreted phospholipases in Gram-negative bacteria.
Keywords: Fusobacterium; autotransporter; chemical biology; colorectal cancer; host-pathogen interaction; phospholipase A; protein secretion.
© 2017 by The American Society for Biochemistry and Molecular Biology, Inc.
Conflict of interest statement
The authors declare that they have no conflicts of interest with the contents of this article
Figures




References
-
- Signat B., Roques C., Poulet P., and Duffaut D. (2011) Fusobacterium nucleatum in periodontal health and disease. Curr. Issues Mol. Biol. 13, 25–36 - PubMed
-
- Abed J., Emgård J. E., Zamir G., Faroja M., Almogy G., Grenov A., Sol A., Naor R., Pikarsky E., Atlan K. A., Mellul A., Chaushu S., Manson A. L., Earl A. M., Ou N., et al. (2016) Fap2 mediates Fusobacterium nucleatum colorectal adenocarcinoma enrichment by binding to tumor-expressed Gal-GalNAc. Cell Host Microbe 20, 215–225 - PMC - PubMed
-
- Dahya V., Patel J., Wheeler M., and Ketsela G. (2015) Fusobacterium nucleatum endocarditis presenting as liver and brain abscesses in an immunocompetent patient. Am. J. Med. Sci. 349, 284–285 - PubMed
-
- Rashidi A., Tahhan S. G., Cohee M. W., and Goodman B. M. (2012) Fusobacterium nucleatum infection mimicking metastatic cancer. Indian J. Gastroenterol. 31, 198–200 - PubMed
MeSH terms
Substances
Associated data
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

