Early Right Ventricular Assist Device Use in Patients Undergoing Continuous-Flow Left Ventricular Assist Device Implantation: Incidence and Risk Factors From the Interagency Registry for Mechanically Assisted Circulatory Support
- PMID: 29021348
- PMCID: PMC5717751
- DOI: 10.1161/CIRCHEARTFAILURE.117.003863
Early Right Ventricular Assist Device Use in Patients Undergoing Continuous-Flow Left Ventricular Assist Device Implantation: Incidence and Risk Factors From the Interagency Registry for Mechanically Assisted Circulatory Support
Abstract
Background: To investigate preimplant risk factors associated with early right ventricular assist device (RVAD) use in patients undergoing continuous-flow left ventricular assist device (LVAD) surgery.
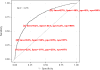
Methods and results: Patients in the Interagency Registry for Mechanically Assisted Circulatory Support who underwent primary continuous-flow-LVAD surgery were examined for concurrent or subsequent RVAD implantation within 14 days of LVAD. Risk factors for RVAD implantation and the combined end point of RVAD or death within 14 days of LVAD were assessed with stepwise logistic regression. We compared survival between patients with and without RVAD using Kaplan-Meier method and Cox proportional hazards modeling. Of 9976 patients undergoing continuous-flow-LVAD implantation, 386 patients (3.9%) required an RVAD within 14 days of LVAD surgery. Preimplant characteristics associated with RVAD use included interagency registry for mechanically assisted circulatory support patient profiles 1 and 2, the need for preoperative extracorporeal membrane oxygenation or renal replacement therapy, severe preimplant tricuspid regurgitation, history of cardiac surgery, and concomitant procedures other than tricuspid valve repair at the time of LVAD. Hemodynamic determinants included elevated right atrial pressure, reduced pulmonary artery pulse pressure, and reduced stroke volume. The final model demonstrated good performance for both RVAD implant (area under the curve, 0.78) and the combined end point of RVAD or death within 14 days (area under the curve, 0.73). Compared with patients receiving an isolated LVAD, patients requiring RVAD had decreased 1- and 6-month survival: 78.1% versus 95.8% and 63.6% versus 87.9%, respectively (P<0.0001 for both).
Conclusions: The need for RVAD implantation after LVAD is associated with indices of global illness severity, markers of end-organ dysfunction, and profiles of hemodynamic instability.
Keywords: atrial pressure; blood pressure; heart; humans; risk factors.
© 2017 American Heart Association, Inc.
Figures

References
-
- Kirklin JK, Naftel DC, Pagani FD, Kormos RL, Stevenson LW, Blume ED, Myers SL, Miller MA, Baldwin JT, Young JB. Seventh INTERMACS annual report: 15,000 patients and counting. J Heart Lung Transplant. 2015;34:1495–1504. - PubMed
-
- Kormos RL, Teuteberg JJ, Pagani FD, Russell SD, John R, Miller LW, Massey T, Milano CA, Moazami N, Sundareswaran KS, Farrar DJ. Right ventricular failure in patients with the HeartMate II continuous-flow left ventricular assist device: incidence, risk factors, and effect on outcomes. J Thorac Cardiovasc Surg. 2010;139:1316–24. - PubMed
-
- Aissaoui N, Morshuis M, Schoenbrodt M, Hakim Meibodi K, Kizner L, Borgermann J, Gummert J. Temporary right ventricular mechanical circulatory support for the management of right ventricular failure in critically ill patients. J Thorac Cardiovasc Surg. 2013;146:186–91. - PubMed
-
- Fitzpatrick JR, 3rd, Frederick JR, Hiesinger W, Hsu VM, McCormick RC, Kozin ED, Laporte CM, O'Hara ML, Howell E, Dougherty D, Cohen JE, Southerland KW, Howard JL, Paulson EC, Acker MA, Morris RJ, Woo YJ. Early planned institution of biventricular mechanical circulatory support results in improved outcomes compared with delayed conversion of a left ventricular assist device to a biventricular assist device. J Thorac Cardiovasc Surg. 2009;137:971–7. - PMC - PubMed
-
- Takeda K, Naka Y, Yang JA, Uriel N, Colombo PC, Jorde UP, Takayama H. Outcome of unplanned right ventricular assist device support for severe right heart failure after implantable left ventricular assist device insertion. J Heart Lung Transplant. 2014;33:141–8. - PubMed
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

