Beyond Antigenic Match: Possible Agent-Host and Immuno-epidemiological Influences on Influenza Vaccine Effectiveness During the 2015-2016 Season in Canada
- PMID: 29029166
- PMCID: PMC5853508
- DOI: 10.1093/infdis/jix526
Beyond Antigenic Match: Possible Agent-Host and Immuno-epidemiological Influences on Influenza Vaccine Effectiveness During the 2015-2016 Season in Canada
Abstract
Background: Vaccine effectiveness (VE) estimates for 2015-2016 seasonal influenza vaccine are reported from Canada's Sentinel Practitioner Surveillance Network (SPSN). This season was characterized by a delayed 2009 pandemic influenza A(H1N1) virus (A[H1N1]pdm09) epidemic and concurrent influenza B(Victoria) virus activity. Potential influences on VE beyond antigenic match are explored, including viral genomic variation, birth cohort effects, prior vaccination, and epidemic period.
Methods: VE was estimated by a test-negative design comparing the adjusted odds ratio for influenza test positivity among vaccinated compared to unvaccinated participants. Vaccine-virus relatedness was assessed by gene sequencing and hemagglutination inhibition assay.
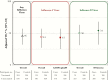
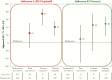
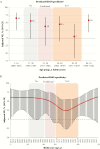
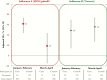
Results: Analyses included 596 influenza A(H1N1)pdm09 and 305 B(Victoria) cases and 926 test-negative controls. A(H1N1)pdm09 viruses were considered antigenically related to vaccine (unchanged since 2009), despite phylogenetic clustering within emerging clade 6B.1. The adjusted VE against A(H1N1)pdm09 was 43% (95% confidence interval [CI], 25%-57%). Compared to other age groups, VE against A(H1N1)pdm09 was lower for adults born during 1957-1976 (25%; 95% CI, -16%-51%). The VE against A(H1N1)pdm09 was also lower for participants consecutively vaccinated during both the current and prior seasons (41%; 95% CI, 18%-57%) than for those vaccinated during the current season only (75%; 95% CI, 45%-88%), and the VE among participants presenting in March-April 2016 (19%; 95% CI, -15%-44%) was lower than that among those presenting during January-February 2016 (62%; 95% CI, 44%-74%). The adjusted VE for B(Victoria) viruses was 54% (95% CI, 32%-68%), despite lineage-level mismatch to B(Yamagata) vaccine. The further variation in VE as observed for A(H1N1)pdm09 was not observed for B(Victoria).
Conclusions: Influenza VE findings may require consideration of other agent-host and immuno-epidemiologic influences on vaccine performance beyond antigenic match, including viral genomic variation, repeat vaccination, birth (immunological) cohort effects, and potential within-season waning of vaccine protection.
Keywords: Influenza; birth cohort effects; hemagglutination inhibition; influenza A subtype; influenza B lineage; influenza vaccine; original antigenic sin; repeat vaccination; sequencing; vaccine effectiveness.
© The Author 2017. Published by Oxford University Press for the Infectious Diseases Society of America.
Figures

Comment in
-
Beyond Antigenic Match: Moving Toward Greater Understanding of Influenza Vaccine Effectiveness.J Infect Dis. 2017 Dec 19;216(12):1477-1480. doi: 10.1093/infdis/jix527. J Infect Dis. 2017. PMID: 29029175 No abstract available.
References
-
- Public Health Agency of Canada. FluWatch report: August 14 to August 27, 2016 (weeks 33–34) http://healthycanadians.gc.ca/publications/diseases-conditions-maladies-.... Accessed 14 August 2017.
-
- United States Centers for Disease Control and Prevention. Fluview 2015–2016 influenza season week 39 ending October 1, 2016 https://www.cdc.gov/flu/weekly/weeklyarchives2015-2016/Week39.htm. Accessed 14 August 2017.
-
- Public Health Agency of Canada. FluWatch report: 27 July to 9 August, 2014 (weeks 31 & 32) http://publications.gc.ca/collections/collection-2014/aspc-phac/HP58-1-2.... Accessed 14 August 2017.
-
- Skowronski DM, Chambers C, Sabaiduc S et al. Integrated sentinel surveillance linking genetic, antigenic, and epidemiologic monitoring of influenza vaccine-virus relatedness and effectiveness during the 2013-2014 influenza season. J Infect Dis 2015; 212:726–39. - PubMed
-
- Epperson S, Blanton L, Kniss K et al. Influenza activity—United States, 2013–14 season and composition of the 2014–15 influenza vaccines. MMWR Morb Mortal Wkly Rep 2014; 63:483–90. https://www.cdc.gov/mmwr/pdf/wk/mm6322.pdf. Accessed 14 August 2017. - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

