Development of mesenchymal subtype gene signature for clinical application in gastric cancer
- PMID: 29029513
- PMCID: PMC5630413
- DOI: 10.18632/oncotarget.19985
Development of mesenchymal subtype gene signature for clinical application in gastric cancer
Abstract
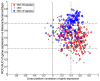
Previously, in the Asian Cancer Research Group (ACRG) project, we defined four distinct molecular subtypes in gastric cancer (GC). Mesenchymal (microsatellite stable with epithelial-to-mesenchymal transition phenotype, MSS/EMT) tumors showed the worst prognosis among all the subtypes. To develop a gene signature for predicting mesenchymal subtype GC, we conducted gene expression profiling using a NanoString assay in 70 ACRG specimens. The gene signature was validated in an independent set obtained from the prospective Adjuvant chemoRadioTherapy In Stomach Tumor (ARTIST) trial. The association between the mesenchymal subtype and survival was investigated. After cross-platform concordance test performed in 70 ACRG specimens, a 71-gene MSS/EMT signature was obtained. In the validation set, the gene signature predicted that 20 of 73 (27%) patients had mesenchymal tumors. Patients with mesenchymal subtype had diffuse GC, poorly-differentiated or signet ring cell carcinoma, and were microsatellite stable. The estimated hazard ratio for survival in patients with mesenchymal GC compared to those with non-mesenchymal tumors was 2.262 (95% confidence interval, 1.410 to 3.636; P=0.001). The survival difference remained significant when the subtypes were analyzed according to clinical prognostic parameters. This study suggested that the NanoString-based 71-gene signature for mesenchymal subtype is a strong predictor of the outcome in patients with GC.
Keywords: gene signature; genomics; mesenchymal; stomach neoplasm; subtypes.
Figures


References
-
- Torre LA, Bray F, Siegel RL, Ferlay J, Lortet-Tieulent J, Jemal A. Global cancer statistics, 2012. CA Cancer J Clin. 2015;65:87–108. - PubMed
-
- Bang YJ, Van Cutsem E, Feyereislova A, Chung HC, Shen L, Sawaki A, Lordick F, Ohtsu A, Omuro Y, Satoh T, Aprile G, Kulikov E, Hill J, et al. Trastuzumab in combination with chemotherapy versus chemotherapy alone for treatment of HER2-positive advanced gastric or gastro-oesophageal junction cancer (ToGA): a phase 3, open-label, randomised controlled trial. Lancet. 2010;376:687–697. - PubMed
-
- Fuchs CS, Tomasek J, Yong CJ, Dumitru F, Passalacqua R, Goswami C, Safran H, dos Santos LV, Aprile G, Ferry DR, Melichar B, Tehfe M, Topuzov E, et al. Ramucirumab monotherapy for previously treated advanced gastric or gastro-oesophageal junction adenocarcinoma (REGARD): an international, randomised, multicentre, placebo-controlled, phase 3 trial. Lancet. 2014;383:31–39. - PubMed
-
- Wilke H, Muro K, Van Cutsem E, Oh SC, Bodoky G, Shimada Y, Hironaka S, Sugimoto N, Lipatov O, Kim TY, Cunningham D, Rougier P, Komatsu Y, et al. Ramucirumab plus paclitaxel versus placebo plus paclitaxel in patients with previously treated advanced gastric or gastro-oesophageal junction adenocarcinoma (RAINBOW): a double-blind, randomised phase 3 trial. Lancet Oncol. 2014;15:1224–1235. - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

