Epigenetic Activation of Pro-angiogenic Signaling Pathways in Human Endothelial Progenitors Increases Vasculogenesis
- PMID: 29033304
- PMCID: PMC5830028
- DOI: 10.1016/j.stemcr.2017.09.009
Epigenetic Activation of Pro-angiogenic Signaling Pathways in Human Endothelial Progenitors Increases Vasculogenesis
Abstract
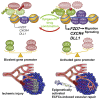
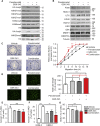
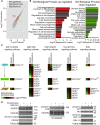
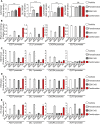
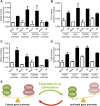
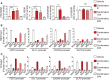
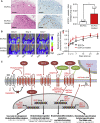
Human endothelial colony-forming cells (ECFCs) represent a promising source of adult stem cells for vascular repair, yet their regenerative capacity is limited. Here, we set out to understand the molecular mechanism restricting the repair function of ECFCs. We found that key pro-angiogenic pathways are repressed in ECFCs due to the presence of bivalent (H3K27me3/H3K4me3) epigenetic marks, which decreases the cells' regenerative potential. Importantly, ex vivo treatment with a combination of epigenetic drugs that resolves bivalent marks toward the transcriptionally active H3K4me3 state leads to the simultaneous activation of multiple pro-angiogenic signaling pathways (VEGFR, CXCR4, WNT, NOTCH, SHH). This in turn results in improved capacity of ECFCs to form capillary-like networks in vitro and in vivo. Furthermore, restoration of perfusion is accelerated upon transplantation of drug-treated ECFCs in a model of hindlimb ischemia. Thus, ex vivo treatment with epigenetic drugs increases the vascular repair properties of ECFCs through transient activation of pro-angiogenic signaling pathways.
Keywords: ECFCs; EZH2; UTX; angiogenesis; bivalent genes; epigenetics; hindlimb ischemia; human endothelial progenitors; pro-angiogenic pathway; vasculogenesis.
Copyright © 2017 The Authors. Published by Elsevier Inc. All rights reserved.
Figures
References
-
- Alphonse R.S., Vadivel A., Fung M., Shelley W.C., Critser P.J., Ionescu L., O'Reilly M., Ohls R.K., McConaghy S., Eaton F. Existence, functional impairment, and lung repair potential of endothelial colony-forming cells in oxygen-induced arrested alveolar growth. Circulation. 2014;129:2144–2157. - PMC - PubMed
-
- Au P., Daheron L.M., Duda D.G., Cohen K.S., Tyrrell J.A., Lanning R.M., Fukumura D., Scadden D.T., Jain R.K. Differential in vivo potential of endothelial progenitor cells from human umbilical cord blood and adult peripheral blood to form functional long-lasting vessels. Blood. 2008;111:1302–1305. - PMC - PubMed
-
- Benyoucef A., Brand M. Epigenetic gene regulation and stem cell function. In: Huang S., Litt M.D., Blakey C.A., editors. Epigenetic Gene Expression and Regulation. Elsevier; 2015. pp. 149–181.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

