Lack of myeloid Fatp1 increases atherosclerotic lesion size in Ldlr-/- mice
- PMID: 29035781
- PMCID: PMC5705203
- DOI: 10.1016/j.atherosclerosis.2017.10.009
Lack of myeloid Fatp1 increases atherosclerotic lesion size in Ldlr-/- mice
Abstract
Background and aims: Altered metabolism is an important regulator of macrophage (MΦ) phenotype, which contributes to inflammatory diseases such as atherosclerosis. Broadly, pro-inflammatory, classically-activated MΦs (CAM) are glycolytic while alternatively-activated MΦs (AAM) oxidize fatty acids, although overlap exists. We previously demonstrated that MΦ fatty acid transport protein 1 (FATP1, Slc27a1) was necessary to maintain the oxidative and anti-inflammatory AAM phenotype in vivo in a model of diet-induced obesity. The aim of this study was to examine how MΦ metabolic reprogramming through FATP1 ablation affects the process of atherogenesis. We hypothesized that FATP1 limits MΦ-mediated inflammation during atherogenesis. Thus, mice lacking MΦ Fatp1 would display elevated formation of atherosclerotic lesions in a mouse model lacking the low-density lipoprotein (LDL) receptor (Ldlr-/-).
Methods: We transplanted bone marrow collected from Fatp1+/+ or Fatp1-/- mice into Ldlr-/- mice and fed chimeric mice a Western diet for 12 weeks. Body weight, blood glucose, and plasma lipids were measured. Aortic sinus and aorta lesions were quantified. Atherosclerotic plaque composition, oxidative stress, and inflammation were analyzed histologically.
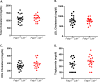
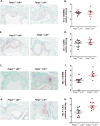
Results: Compared to Fatp1+/+Ldlr-/- mice, Fatp1-/-Ldlr-/- mice exhibited significantly larger lesion area and elevated oxidative stress and inflammation in the atherosclerotic plaque. Macrophage and smooth muscle cell content did not differ by Fatp1 genotype. There were no significant systemic alterations in LDL, high-density lipoprotein (HDL), total cholesterol, or triacylglyceride, suggesting that the effect was local to the cells of the vessel microenvironment in a Fatp1-dependent manner.
Conclusions: MΦ Fatp1 limits atherogenesis and may be a viable target to metabolically reprogram MΦs.
Keywords: FATP1; Fatty acid transport protein; LDL receptor; Macrophage; Metabolic reprogram.
Copyright © 2017 Elsevier B.V. All rights reserved.
Conflict of interest statement
The authors declared they do not have anything to disclose regarding conflict of interest with respect to this manuscript.
Figures


References
-
- Schaftenaar F, et al. Atherosclerosis: the interplay between lipids and immune cells. Curr Opin Lipidol. 2016;27(3):209–15. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials

