Integrated Process Modeling-A Process Validation Life Cycle Companion
- PMID: 29039771
- PMCID: PMC5746753
- DOI: 10.3390/bioengineering4040086
Integrated Process Modeling-A Process Validation Life Cycle Companion
Abstract
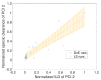
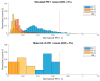
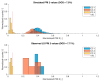
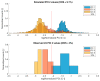
During the regulatory requested process validation of pharmaceutical manufacturing processes, companies aim to identify, control, and continuously monitor process variation and its impact on critical quality attributes (CQAs) of the final product. It is difficult to directly connect the impact of single process parameters (PPs) to final product CQAs, especially in biopharmaceutical process development and production, where multiple unit operations are stacked together and interact with each other. Therefore, we want to present the application of Monte Carlo (MC) simulation using an integrated process model (IPM) that enables estimation of process capability even in early stages of process validation. Once the IPM is established, its capability in risk and criticality assessment is furthermore demonstrated. IPMs can be used to enable holistic production control strategies that take interactions of process parameters of multiple unit operations into account. Moreover, IPMs can be trained with development data, refined with qualification runs, and maintained with routine manufacturing data which underlines the lifecycle concept. These applications will be shown by means of a process characterization study recently conducted at a world-leading contract manufacturing organization (CMO). The new IPM methodology therefore allows anticipation of out of specification (OOS) events, identify critical process parameters, and take risk-based decisions on counteractions that increase process robustness and decrease the likelihood of OOS events.
Keywords: Monte Carlo simulation; biopharmaceutical manufacturing; holistic process model; predict out of specification events; process characterization study; process validation.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


References
-
- Process Validation: General Principles and Practices. U.S. Department of Health and Human Services; Washington, DC, USA: 2011.
-
- Guideline I.H.T. Pharmaceutical Development Q8 (R2) Curr. Step. 2009;4:1–24.
-
- Guideline I.H.T. Quality risk management, Q9. Curr. Step. 2005;4:408.
-
- Katz P., Campbell C. FDA 2011 process validation guidance: Process validation revisited. J. GXP Compliance. 2012;16:18. - PubMed
-
- Peterson J.J., Lief K. The ICH Q8 definition of design space: A comparison of the overlapping means and the bayesian predictive approaches. Stat. Biopharm. Res. 2010;2:249–259. doi: 10.1198/sbr.2009.08065. - DOI
LinkOut - more resources
Full Text Sources
Other Literature Sources

