Sperm Release at Spermiation Is Regulated by Changes in the Organization of Actin- and Microtubule-Based Cytoskeletons at the Apical Ectoplasmic Specialization-A Study Using the Adjudin Model
- PMID: 29040437
- PMCID: PMC5711386
- DOI: 10.1210/en.2017-00660
Sperm Release at Spermiation Is Regulated by Changes in the Organization of Actin- and Microtubule-Based Cytoskeletons at the Apical Ectoplasmic Specialization-A Study Using the Adjudin Model
Abstract
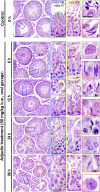
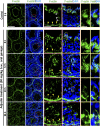
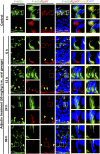
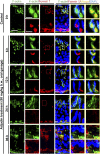
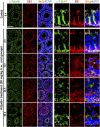
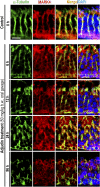
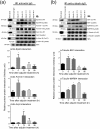
The mechanism that regulates sperm release at spermiation is unknown. Herein, we used an animal model wherein rats were treated with adjudin, 1-(2,4-dichlorobenzyl)-1H-indazole-3-carbohydrazide, via oral gavage to induce premature release of elongating/elongated spermatids, followed by round spermatids and spermatocytes. Spermatid release mimicking spermiation occurred within 6 to 12 hours following adjudin treatment and, by 96 hours, virtually all tubules were devoid of elongating/elongated spermatids. Using this model, we tracked the organization of F-actin and microtubules (MTs) by immunofluorescence microscopy, and the association of actin or MT regulatory proteins that either promote or demolish cytoskeletal integrity through changes in the organization of actin microfilaments or MTs by coimmunoprecipitation. Adjudin treatment induced an increase in the association of (1) epidermal growth factor receptor pathway substrate 8 (an actin barbed-end capping and bundling protein) or formin 1 (an actin nucleator) with actin and (2) end-binding protein 1 (an MT stabilizing protein) with MT shortly after adjudin exposure (at 6 hours), in an attempt to maintain spermatid adhesion to the Sertoli cell at the apical ectoplasmic specialization (ES). However, this was followed by a considerable decline of their steady-state protein levels, replacing with an increase in association of (1) actin-related protein 3 (a branched actin nucleator that converts actin filaments into a branched/unbundled network) with actin and (2) MT affinity-regulating kinase 4 (an MT destabilizing protein kinase) with MTs by 12 hours after adjudin treatment. These latter changes thus promoted actin and MT disorganization, leading to apical ES disruption and the release of elongating/elongated spermatids, mimicking spermiation. In summary, spermiation is a cytoskeletal-dependent event, involving regulatory proteins that modify cytoskeletal organization.
Copyright © 2017 Endocrine Society.
Figures

Similar articles
-
Coordination of Actin- and Microtubule-Based Cytoskeletons Supports Transport of Spermatids and Residual Bodies/Phagosomes During Spermatogenesis in the Rat Testis.Endocrinology. 2016 Apr;157(4):1644-59. doi: 10.1210/en.2015-1962. Epub 2016 Feb 19. Endocrinology. 2016. PMID: 26894662 Free PMC article.
-
Breast cancer resistance protein regulates apical ectoplasmic specialization dynamics stage specifically in the rat testis.Am J Physiol Endocrinol Metab. 2013 Apr 1;304(7):E757-69. doi: 10.1152/ajpendo.00645.2012. Epub 2013 Feb 12. Am J Physiol Endocrinol Metab. 2013. PMID: 23403943 Free PMC article.
-
p-FAK-Tyr(397) regulates spermatid adhesion in the rat testis via its effects on F-actin organization at the ectoplasmic specialization.Am J Physiol Endocrinol Metab. 2013 Sep 15;305(6):E687-99. doi: 10.1152/ajpendo.00254.2013. Epub 2013 Jul 23. Am J Physiol Endocrinol Metab. 2013. PMID: 23880313 Free PMC article.
-
Transport of germ cells across the seminiferous epithelium during spermatogenesis-the involvement of both actin- and microtubule-based cytoskeletons.Tissue Barriers. 2016 Nov 28;4(4):e1265042. doi: 10.1080/21688370.2016.1265042. eCollection 2016. Tissue Barriers. 2016. PMID: 28123928 Free PMC article. Review.
-
Tubulobulbar complex: cytoskeletal remodeling to release spermatozoa.Reprod Biol Endocrinol. 2012 Apr 17;10:27. doi: 10.1186/1477-7827-10-27. Reprod Biol Endocrinol. 2012. PMID: 22510523 Free PMC article. Review.
Cited by
-
Bone morphogenetic protein 4 inhibits rat stem/progenitor Leydig cell development and regeneration via SMAD-dependent and SMAD-independent signaling.Cell Death Dis. 2022 Dec 13;13(12):1039. doi: 10.1038/s41419-022-05471-8. Cell Death Dis. 2022. PMID: 36513649 Free PMC article.
-
FYN regulates cell adhesion at the blood-testis barrier and the apical ectoplasmic specialization via its effect on Arp3 in the mouse testis.Front Immunol. 2022 Aug 9;13:915274. doi: 10.3389/fimmu.2022.915274. eCollection 2022. Front Immunol. 2022. PMID: 36016954 Free PMC article.
-
Regulation of blood-testis barrier dynamics by the mTORC1/rpS6 signaling complex: An in vitro study.Asian J Androl. 2019 Jul-Aug;21(4):365-375. doi: 10.4103/aja.aja_126_18. Asian J Androl. 2019. PMID: 30829292 Free PMC article.
-
Endogenously produced LG3/4/5-peptide protects testes against toxicant-induced injury.Cell Death Dis. 2020 Jun 8;11(6):436. doi: 10.1038/s41419-020-2608-8. Cell Death Dis. 2020. PMID: 32513914 Free PMC article.
-
Spermiation: Insights from Studies on the Adjudin Model.Adv Exp Med Biol. 2021;1288:241-254. doi: 10.1007/978-3-030-77779-1_12. Adv Exp Med Biol. 2021. PMID: 34453740 Review.
References
-
- Cheng CY, Mruk DD. Biochemistry of Sertoli cell/germ cell junctions, germ cell transport, and spermiation in the seminiferous epithelium In: Griswold MD, ed. Sertoli Cell Biology. 2nd ed. Amsterdam: Elsevier; 2015:333–383.
-
- Amann RP. The cycle of the seminiferous epithelium in humans: a need to revisit? J Androl. 2008;29(5):469–487. - PubMed
-
- Hess RA, Renato de Franca L. Spermatogenesis and cycle of the seminiferous epithelium. Adv Exp Med Biol. 2008;636:1–15. - PubMed
-
- Vogl AW, Vaid KS, Guttman JA. The Sertoli cell cytoskeleton. Adv Exp Med Biol. 2008;636:186–211. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

