Adenylyl cyclase 5 deficiency reduces renal cyclic AMP and cyst growth in an orthologous mouse model of polycystic kidney disease
- PMID: 29042084
- PMCID: PMC5794572
- DOI: 10.1016/j.kint.2017.08.005
Adenylyl cyclase 5 deficiency reduces renal cyclic AMP and cyst growth in an orthologous mouse model of polycystic kidney disease
Abstract
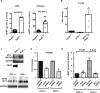
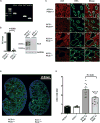
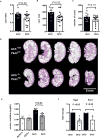
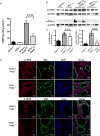
Cyclic AMP promotes cyst growth in polycystic kidney disease (PKD) by stimulating cell proliferation and fluid secretion. Previously, we showed that the primary cilium of renal epithelial cells contains a cAMP regulatory complex comprising adenylyl cyclases 5 and 6 (AC5/6), polycystin-2, A-kinase anchoring protein 150, protein kinase A, and phosphodiesterase 4C. In Kif3a mutant cells that lack primary cilia, the formation of this regulatory complex is disrupted and cAMP levels are increased. Inhibition of AC5 reduces cAMP levels in Kif3a mutant cells, suggesting that AC5 may mediate the increase in cAMP in PKD. Here, we examined the role of AC5 in an orthologous mouse model of PKD caused by kidney-specific ablation of Pkd2. Knockdown of AC5 with siRNA attenuated the increase in cAMP levels in Pkd2-deficient renal epithelial cells. Levels of cAMP and AC5 mRNA transcripts were elevated in the kidneys of mice with collecting duct-specific ablation of Pkd2. Compared with Pkd2 single mutant mice, AC5/Pkd2 double mutant mice had less kidney enlargement, lower cyst index, reduced kidney injury, and improved kidney function. Importantly, cAMP levels and cAMP-dependent signaling were reduced in the kidneys of AC5/Pkd2 double mutant compared to the kidneys of Pkd2 single mutant mice. Additionally, we localized endogenous AC5 in the primary cilium of renal epithelial cells and showed that ablation of AC5 reduced ciliary elongation in the kidneys of Pkd2 mutant mice. Thus, AC5 contributes importantly to increased renal cAMP levels and cyst growth in Pkd2 mutant mice, and inhibition of AC5 may be beneficial in the treatment of PKD.
Keywords: adenylyl cyclase; cilia; cyclic AMP; knockout mice; polycystic kidney disease; polycystin-2.
Copyright © 2017 International Society of Nephrology. Published by Elsevier Inc. All rights reserved.
Figures
Similar articles
-
Adenylyl cyclase 5 links changes in calcium homeostasis to cAMP-dependent cyst growth in polycystic liver disease.J Hepatol. 2017 Mar;66(3):571-580. doi: 10.1016/j.jhep.2016.10.032. Epub 2016 Nov 5. J Hepatol. 2017. PMID: 27826057 Free PMC article.
-
Adenylyl cyclase 6 deficiency ameliorates polycystic kidney disease.J Am Soc Nephrol. 2014 Feb;25(2):232-7. doi: 10.1681/ASN.2013010077. Epub 2013 Oct 24. J Am Soc Nephrol. 2014. PMID: 24158982 Free PMC article.
-
Polycystin-2 and phosphodiesterase 4C are components of a ciliary A-kinase anchoring protein complex that is disrupted in cystic kidney diseases.Proc Natl Acad Sci U S A. 2011 Jun 28;108(26):10679-84. doi: 10.1073/pnas.1016214108. Epub 2011 Jun 13. Proc Natl Acad Sci U S A. 2011. PMID: 21670265 Free PMC article.
-
Cilia and polycystic kidney disease.Semin Cell Dev Biol. 2021 Feb;110:139-148. doi: 10.1016/j.semcdb.2020.05.003. Epub 2020 May 28. Semin Cell Dev Biol. 2021. PMID: 32475690 Review.
-
The genetics and physiology of polycystic kidney disease.Semin Nephrol. 2001 Mar;21(2):107-23. doi: 10.1053/snep.2001.20929. Semin Nephrol. 2001. PMID: 11245774 Review.
Cited by
-
Direct visualization of cAMP signaling in primary cilia reveals up-regulation of ciliary GPCR activity following Hedgehog activation.Proc Natl Acad Sci U S A. 2019 Jun 11;116(24):12066-12071. doi: 10.1073/pnas.1819730116. Epub 2019 May 29. Proc Natl Acad Sci U S A. 2019. PMID: 31142652 Free PMC article.
-
A methionine-Mettl3-N6-methyladenosine axis promotes polycystic kidney disease.Cell Metab. 2021 Jun 1;33(6):1234-1247.e7. doi: 10.1016/j.cmet.2021.03.024. Epub 2021 Apr 13. Cell Metab. 2021. PMID: 33852874 Free PMC article.
-
Tulp3 Regulates Renal Cystogenesis by Trafficking of Cystoproteins to Cilia.Curr Biol. 2019 Mar 4;29(5):790-802.e5. doi: 10.1016/j.cub.2019.01.047. Epub 2019 Feb 21. Curr Biol. 2019. PMID: 30799239 Free PMC article.
-
Role of transcription factor hepatocyte nuclear factor-1β in polycystic kidney disease.Cell Signal. 2020 Jul;71:109568. doi: 10.1016/j.cellsig.2020.109568. Epub 2020 Feb 14. Cell Signal. 2020. PMID: 32068086 Free PMC article. Review.
-
Insights Into the Molecular Mechanisms of Polycystic Kidney Diseases.Front Physiol. 2021 Sep 8;12:693130. doi: 10.3389/fphys.2021.693130. eCollection 2021. Front Physiol. 2021. PMID: 34566674 Free PMC article. Review.
References
-
- Igarashi P, Somlo S. Genetics and pathogenesis of polycystic kidney disease. J Am Soc Nephrol. 2002;13:2384–2398. - PubMed
-
- Pazour GJ, San Agustin JT, Follit JA, et al. Polycystin-2 localizes to kidney cilia and the ciliary level is elevated in orpk mice with polycystic kidney disease. Curr Biol. 2002;12:R378–R380. - PubMed
-
- Yoder BK, Hou X, Guay-Woodford LM. The polycystic kidney disease proteins, polycystin-1, polycystin-2, polaris, and cystin, are co-localized in renal cilia. J Am Soc Nephrol. 2002;13:2508–2516. - PubMed
-
- Nauli SM, Alenghat FJ, Luo Y, et al. Polycystins 1 and 2 mediate mechanosensation in the primary cilium of kidney cells. Nat Genet. 2003;33:129–137. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

