Selectivity of coronaridine congeners at nicotinic acetylcholine receptors and inhibitory activity on mouse medial habenula
- PMID: 29042244
- PMCID: PMC5903430
- DOI: 10.1016/j.biocel.2017.10.006
Selectivity of coronaridine congeners at nicotinic acetylcholine receptors and inhibitory activity on mouse medial habenula
Abstract
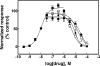
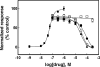
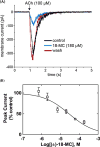
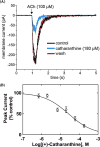
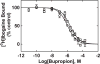
The inhibitory activity of coronaridine congeners on human (h) α4β2 and α7 nicotinic acetylcholine receptors (AChRs) is determined by Ca2+ influx assays, whereas their effects on neurons in the ventral inferior (VI) aspect of the mouse medial habenula (MHb) are determined by patch-clamp recordings. The Ca2+ influx results clearly establish that coronaridine congeners inhibit hα3β4 AChRs with higher selectivity compared to hα4β2 and hα7 subtypes, and with the following potency sequence, for hα4β2: (±)-18-methoxycoronaridine [(±)-18-MC]>(+)-catharanthine>(±)-18-methylaminocoronaridine [(±)-18-MAC] ∼ (±)-18-hydroxycoronaridine [(±)-18-HC]; and for hα7: (+)-catharanthine>(±)-18-MC>(±)-18-HC>(±)-18-MAC. Interestingly, the inhibitory potency of (+)-catharanthine (27±4μM) and (±)-18-MC (28±6μM) on MHb (VI) neurons was lower than that observed on hα3β4 AChRs, suggesting that these compounds inhibit a variety of endogenous α3β4* AChRs. In addition, the interaction of bupropion with (-)-ibogaine sites on hα3β4 AChRs is tested by [3H]ibogaine competition binding experiments. The results indicate that bupropion binds to ibogaine sites at desensitized hα3β4 AChRs with 2-fold higher affinity than at resting receptors, suggesting that these compounds share the same binding sites. In conclusion, coronaridine congeners inhibit hα3β4 AChRs with higher selectivity compared to other AChRs, by interacting with the bupropion (luminal) site. Coronaridine congeners also inhibit α3β4*AChRs expressed in MHb (VI) neurons, supporting the notion that these receptors are important endogenous targets for their anti-addictive activities.
Keywords: (+)-Catharanthine; 18-Methoxycoronaridine; Brain slices; Coronaridine congeners; Medial habenula; Nicotinic acetylcholine receptor.
Copyright © 2017 Elsevier Ltd. All rights reserved.
Figures

Similar articles
-
Selectivity of (±)-citalopram at nicotinic acetylcholine receptors and different inhibitory mechanisms between habenular α3β4* and α9α10 subtypes.Neurochem Int. 2019 Dec;131:104552. doi: 10.1016/j.neuint.2019.104552. Epub 2019 Sep 20. Neurochem Int. 2019. PMID: 31545995 Free PMC article.
-
Coronaridine congeners inhibit human α3β4 nicotinic acetylcholine receptors by interacting with luminal and non-luminal sites.Int J Biochem Cell Biol. 2015 Aug;65:81-90. doi: 10.1016/j.biocel.2015.05.015. Epub 2015 May 27. Int J Biochem Cell Biol. 2015. PMID: 26022277
-
Structure-activity relationship of ibogaine analogs interacting with nicotinic acetylcholine receptors in different conformational states.Int J Biochem Cell Biol. 2011 Sep;43(9):1330-9. doi: 10.1016/j.biocel.2011.05.011. Epub 2011 May 27. Int J Biochem Cell Biol. 2011. PMID: 21642011
-
Is the inhibition of nicotinic acetylcholine receptors by bupropion involved in its clinical actions?Int J Biochem Cell Biol. 2009 Nov;41(11):2098-108. doi: 10.1016/j.biocel.2009.05.015. Epub 2009 Jun 2. Int J Biochem Cell Biol. 2009. PMID: 19497387 Review.
-
DARK Classics in Chemical Neuroscience: Ibogaine.ACS Chem Neurosci. 2018 Oct 17;9(10):2475-2483. doi: 10.1021/acschemneuro.8b00294. Epub 2018 Oct 9. ACS Chem Neurosci. 2018. PMID: 30216039 Review.
Cited by
-
Selectivity of (±)-citalopram at nicotinic acetylcholine receptors and different inhibitory mechanisms between habenular α3β4* and α9α10 subtypes.Neurochem Int. 2019 Dec;131:104552. doi: 10.1016/j.neuint.2019.104552. Epub 2019 Sep 20. Neurochem Int. 2019. PMID: 31545995 Free PMC article.
-
What We Have Gained from Ibogaine: α3β4 Nicotinic Acetylcholine Receptor Inhibitors as Treatments for Substance Use Disorders.J Med Chem. 2023 Jan 12;66(1):107-121. doi: 10.1021/acs.jmedchem.2c01562. Epub 2022 Nov 28. J Med Chem. 2023. PMID: 36440853 Free PMC article. Review.
-
(+)-Catharanthine potentiates the GABAA receptor by binding to a transmembrane site at the β(+)/α(-) interface near the TM2-TM3 loop.Biochem Pharmacol. 2022 May;199:114993. doi: 10.1016/j.bcp.2022.114993. Epub 2022 Mar 15. Biochem Pharmacol. 2022. PMID: 35304861 Free PMC article.
-
Is the Antidepressant Activity of Selective Serotonin Reuptake Inhibitors Mediated by Nicotinic Acetylcholine Receptors?Molecules. 2021 Apr 8;26(8):2149. doi: 10.3390/molecules26082149. Molecules. 2021. PMID: 33917953 Free PMC article. Review.
-
Tabernanthalog and ibogainalog inhibit the α7 and α9α10 nicotinic acetylcholine receptors via different mechanisms and with higher potency than the GABAA receptor and CaV2.2 channel.Biochem Pharmacol. 2024 May;223:116183. doi: 10.1016/j.bcp.2024.116183. Epub 2024 Apr 3. Biochem Pharmacol. 2024. PMID: 38580167 Free PMC article.
References
-
- Alper KR, Lotsof HS, Kaplan CD. The ibogaine medical subculture. J Ethnopharmacol. 2008;115:9–24. - PubMed
-
- Arias HR, Feuerbach D, Targowska-Duda KM, Russell M, Jozwiak K. Interaction of selective serotonin reuptake inhibitors with neuronal nicotinic acetylcholine receptors. Biochemistry. 2010c;49:5734–5742. - PubMed
-
- Arias HR, Targowska-Duda KM, Feuerbach D, Sullivan CJ, Maciejewski R, Jozwiak K. Different interaction between tricyclic antidepressants and mecamylamine with the human α3β4 nicotinic acetylcholine receptor ion channel. Neurochem Int. 2010d;56:642–649. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

