Safety, immunogenicity and efficacy after switching from reference infliximab to biosimilar SB2 compared with continuing reference infliximab and SB2 in patients with rheumatoid arthritis: results of a randomised, double-blind, phase III transition study
- PMID: 29042358
- PMCID: PMC5867419
- DOI: 10.1136/annrheumdis-2017-211741
Safety, immunogenicity and efficacy after switching from reference infliximab to biosimilar SB2 compared with continuing reference infliximab and SB2 in patients with rheumatoid arthritis: results of a randomised, double-blind, phase III transition study
Abstract
Objectives: Efficacy, safety and immunogenicity results from the phase III study of SB2, a biosimilar of reference infliximab (INF), were previously reported through 54 weeks. This transition period compared results in patients with rheumatoid arthritis (RA) who switched from INF to SB2 with those in patients who maintained treatment with INF or SB2.
Methods: Patients with moderate to severe RA despite methotrexate treatment were randomised (1:1) to receive SB2 or INF at weeks 0, 2 and 6 and every 8 weeks thereafter until week 46. At week 54, patients previously receiving INF were rerandomised (1:1) to switch to SB2 (INF/SB2 (n=94)) or to continue on INF (INF/INF (n=101)) up to week 70. Patients previously receiving SB2 continued on SB2 (SB2/SB2 (n=201)) up to week 70. Efficacy, safety and immunogenicity were assessed up to week 78.
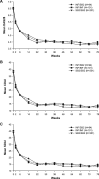
Results: Efficacy was sustained and comparable across treatment groups. American College of Rheumatology (ACR) 20 responses between weeks 54 and 78 ranged from 63.5% to 72.3% with INF/SB2, 66.3%%-69.4% with INF/INF and 65.6%-68.3% with SB2/SB2. Treatment-emergent adverse events during this time occurred in 36.2%, 35.6% and 40.3%, respectively, and infusion-related reactions in 3.2%, 2.0% and 3.5%. Among patients who were negative for antidrug antibodies (ADA) up to week 54, newly developed ADAs were reported in 14.6%, 14.9% and 14.1% of the INF/SB2, INF/INF and SB2/SB2 groups, respectively.
Conclusions: The efficacy, safety and immunogenicity profiles remained comparable among the INF/SB2, INF/INF and SB2/SB2 groups up to week 78, with no treatment-emergent issues or clinically relevant immunogenicity after switching from INF to SB2.
Trial registration number: NCT01936181; EudraCT number: 2012-005733-37.
Keywords: anti-tnf; dmards (biologic); rheumatoid arthritis; tnf-alpha; treatment.
© Article author(s) (or their employer(s) unless otherwise stated in the text of the article) 2018. All rights reserved. No commercial use is permitted unless otherwise expressly granted.
Conflict of interest statement
Competing interests: JSS reports receiving grant/research support from AbbVie, Janssen, MSD, Pfizer, Roche and UCB; consultant for AbbVie, Amgen, AstraZeneca, Astro-Pharma, Celgene, GSK, Janssen, Lilly, Medimmune, MSD, Novartis-Sandoz, Novo Nordisk, Pfizer, Roche, Samsung Bioepis, Sanofi and UCB. J-YC reports receiving grant/research support and consultant fees from Samsung Bioepis. AB reports receiving grant/research support from AbbVie and Samsung Bioepis. NP, JN, IS, ED, RY, MM, WP, HC, KJ-R and AZ report receiving grant/research support from Samsung Bioepis. YL and YHR are employees of Samsung Bioepis and own stocks in Samsung Biologics.
Figures


Similar articles
-
A randomised, double-blind, phase III study comparing SB2, an infliximab biosimilar, to the infliximab reference product Remicade in patients with moderate to severe rheumatoid arthritis despite methotrexate therapy.Ann Rheum Dis. 2017 Jan;76(1):58-64. doi: 10.1136/annrheumdis-2015-207764. Epub 2015 Aug 28. Ann Rheum Dis. 2017. PMID: 26318384 Free PMC article. Clinical Trial.
-
Comparing biosimilar SB2 with reference infliximab after 54 weeks of a double-blind trial: clinical, structural and safety results.Rheumatology (Oxford). 2017 Oct 1;56(10):1771-1779. doi: 10.1093/rheumatology/kex254. Rheumatology (Oxford). 2017. PMID: 28957563 Free PMC article. Clinical Trial.
-
Long-term Efficacy, Safety, and Immunogenicity of the Infliximab (IFX) Biosimilar, PF-06438179/GP1111, in Patients with Rheumatoid Arthritis After Switching from Reference IFX or Continuing Biosimilar Therapy: Week 54-78 Data From a Randomized, Double-Blind, Phase III Trial.BioDrugs. 2020 Apr;34(2):197-207. doi: 10.1007/s40259-019-00403-z. BioDrugs. 2020. PMID: 31939063 Free PMC article. Clinical Trial.
-
SB2: An Infliximab Biosimilar.BioDrugs. 2017 Oct;31(5):461-464. doi: 10.1007/s40259-017-0240-7. BioDrugs. 2017. PMID: 28803431 Review.
-
Efficacy and safety of subcutaneous infliximab versus adalimumab, etanercept and intravenous infliximab in patients with rheumatoid arthritis: a systematic literature review and meta-analysis.Expert Rev Clin Immunol. 2021 Jan;17(1):85-99. doi: 10.1080/1744666X.2020.1858803. Epub 2020 Dec 28. Expert Rev Clin Immunol. 2021. PMID: 33305638
Cited by
-
Systematic review: efficacy and safety of switching patients between reference and biosimilar infliximab.Aliment Pharmacol Ther. 2019 Jan;49(1):31-40. doi: 10.1111/apt.14997. Epub 2018 Nov 8. Aliment Pharmacol Ther. 2019. PMID: 30411382 Free PMC article.
-
Batch-to-Batch Consistency of SB4 and SB2, Etanercept and Infliximab Biosimilars.BioDrugs. 2020 Apr;34(2):225-233. doi: 10.1007/s40259-019-00402-0. BioDrugs. 2020. PMID: 31925703 Free PMC article.
-
The Efficacy, Safety, and Immunogenicity of Switching Between Reference Biopharmaceuticals and Biosimilars: A Systematic Review.Clin Pharmacol Ther. 2020 Oct;108(4):734-755. doi: 10.1002/cpt.1836. Epub 2020 Apr 30. Clin Pharmacol Ther. 2020. PMID: 32236956 Free PMC article.
-
Therapeutic Equivalence of Biosimilar and Reference Biologic Drugs in Rheumatoid Arthritis: A Systematic Review and Meta-analysis.JAMA Netw Open. 2023 May 1;6(5):e2315872. doi: 10.1001/jamanetworkopen.2023.15872. JAMA Netw Open. 2023. PMID: 37234004 Free PMC article.
-
Efficacy and Safety in the Continued Treatment With a Biosimilar Drug in Patients Receiving Infliximab: A Systematic Review in the Context of Decision-Making From a Latin-American Country.Front Pharmacol. 2019 Nov 15;10:1010. doi: 10.3389/fphar.2019.01010. eCollection 2019. Front Pharmacol. 2019. PMID: 31798442 Free PMC article.
References
-
- Nam JL, Ramiro S, Gaujoux-Viala C, et al. . Efficacy of biological disease-modifying antirheumatic drugs: a systematic literature review informing the 2013 update of the EULAR recommendations for the management of rheumatoid arthritis. Ann Rheum Dis 2014;73:516–28. 10.1136/annrheumdis-2013-204577 - DOI - PubMed
-
- Singh SC, Bagnato KM. The economic implications of biosimilars. Am J Manag Care 2015. - PubMed
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

