Wilms tumor protein-dependent transcription of VEGF receptor 2 and hypoxia regulate expression of the testis-promoting gene Sox9 in murine embryonic gonads
- PMID: 29042436
- PMCID: PMC5724013
- DOI: 10.1074/jbc.M117.816751
Wilms tumor protein-dependent transcription of VEGF receptor 2 and hypoxia regulate expression of the testis-promoting gene Sox9 in murine embryonic gonads
Abstract
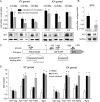
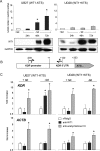
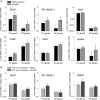
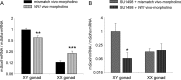
Wilms tumor protein 1 (WT1) has been implicated in the control of several genes in sexual development, but its function in gonad formation is still unclear. Here, we report that WT1 stimulates expression of Kdr, the gene encoding VEGF receptor 2, in murine embryonic gonads. We found that WT1 and KDR are co-expressed in Sertoli cells of the testes and somatic cells of embryonic ovaries. Vivo-morpholino-mediated WT1 knockdown decreased Kdr transcripts in cultured embryonic gonads at multiple developmental stages. Furthermore, WT1 bound to the Kdr promoter in the chromatin of embryonic testes and ovaries. Forced expression of the WT1(-KTS) isoform, which functions as a transcription factor, increased KDR mRNA levels, whereas the WT1(+KTS) isoform, which acts presumably on the post-transcriptional level, did not. ChIP indicated that WT1(-KTS), but not WT1(+KTS), binds to the KDR promoter. Treatment with the KDR tyrosine kinase inhibitor SU1498 or the KDR ligand VEGFA revealed that KDR signaling represses the testis-promoting gene Sox9 in embryonic XX gonads. WT1 knockdown abrogated the stimulatory effect of SU1498-mediated KDR inhibition on Sox9 expression. Exposure to 1% O2 to mimic the low-oxygen conditions in the embryo increased Vegfa expression but did not affect Sox9 mRNA levels in gonadal explants. However, incubation in 1% O2 in the presence of SU1498 significantly reduced Sox9 transcripts in cultured testes and increased Sox9 levels in ovaries. These findings demonstrate that both the local oxygen environment and WT1, which enhances KDR expression, contribute to sex-specific Sox9 expression in developing murine gonads.
Keywords: chromatin immunoprecipitation (ChiP); development; gene regulation; gene silencing; gene transcription; hypoxia; immunohistochemistry; ovary; testis; transcription factor.
© 2017 by The American Society for Biochemistry and Molecular Biology, Inc.
Conflict of interest statement
The authors declare that they have no conflicts of interest with the contents of this article
Figures

Similar articles
-
Transcriptional regulation by the Wilms tumor protein, Wt1, suggests a role of the metalloproteinase Adamts16 in murine genitourinary development.J Biol Chem. 2013 Jun 28;288(26):18811-24. doi: 10.1074/jbc.M113.464644. Epub 2013 May 9. J Biol Chem. 2013. PMID: 23661704 Free PMC article.
-
The Wilms tumor protein WT1 stimulates transcription of the gene encoding insulin-like growth factor binding protein 5 (IGFBP5).Gene. 2017 Jul 1;619:21-29. doi: 10.1016/j.gene.2017.03.037. Epub 2017 Mar 30. Gene. 2017. PMID: 28366834
-
Sry and Sox9 expression during canine gonadal sex determination assayed by quantitative reverse transcription-polymerase chain reaction.Mol Reprod Dev. 2003 Aug;65(4):373-81. doi: 10.1002/mrd.10317. Mol Reprod Dev. 2003. PMID: 12840810
-
Early gonadal development: exploring Wt1 and Sox9 function.Novartis Found Symp. 2002;244:23-31; discussion 31-42, 253-7. Novartis Found Symp. 2002. PMID: 11990794 Review.
-
Genetic control of gonadal differentiation.Baillieres Clin Endocrinol Metab. 1998 Apr;12(1):1-16. doi: 10.1016/s0950-351x(98)80410-2. Baillieres Clin Endocrinol Metab. 1998. PMID: 9890059 Review.
Cited by
-
Involvement of DHH and GLI1 in adrenocortical autograft regeneration in rats.Sci Rep. 2018 Sep 28;8(1):14542. doi: 10.1038/s41598-018-32870-9. Sci Rep. 2018. PMID: 30266964 Free PMC article.
-
Every Beat You Take-The Wilms' Tumor Suppressor WT1 and the Heart.Int J Mol Sci. 2021 Jul 18;22(14):7675. doi: 10.3390/ijms22147675. Int J Mol Sci. 2021. PMID: 34299295 Free PMC article. Review.
-
Implications of the Wilms' Tumor Suppressor Wt1 in Cardiomyocyte Differentiation.Int J Mol Sci. 2021 Apr 21;22(9):4346. doi: 10.3390/ijms22094346. Int J Mol Sci. 2021. PMID: 33919406 Free PMC article.
-
Wt1 haploinsufficiency induces browning of epididymal fat and alleviates metabolic dysfunction in mice on high-fat diet.Diabetologia. 2022 Mar;65(3):528-540. doi: 10.1007/s00125-021-05621-1. Epub 2021 Nov 30. Diabetologia. 2022. PMID: 34846543 Free PMC article.
References
-
- Koopman P., Gubbay J., Vivian N., Goodfellow P., and Lovell-Badge R. (1991) Male development of chromosomally female mice transgenic for Sry. Nature 351, 117–121 - PubMed
-
- Hacker A., Capel B., Goodfellow P., and Lovell-Badge R. (1995) Expression of Sry, the mouse sex determining gene. Development 121, 1603–1614 - PubMed
-
- Albrecht K. H., and Eicher E. M. (2001) Evidence that Sry is expressed in pre-Sertoli cells, and Sertoli and granulosa cells have a common precursor. Dev. Biol. 240, 92–107 - PubMed
-
- Bullejos M., and Koopman P. (2001) Spatially dynamic expression of Sry in mouse genital ridges. Dev. Dyn. 221, 201–205 - PubMed
-
- Chassot A. A., Gregoire E. P., Magliano M., Lavery R., and Chaboissier M. C. (2008) Genetics of ovarian differentiation: Rspo1, a major player. Sex. Dev. 2, 219–227 - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

