The matricellular protein TSP1 promotes human and mouse endothelial cell senescence through CD47 and Nox1
- PMID: 29042481
- PMCID: PMC5679204
- DOI: 10.1126/scisignal.aaj1784
The matricellular protein TSP1 promotes human and mouse endothelial cell senescence through CD47 and Nox1
Abstract
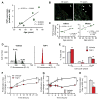
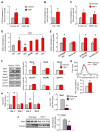
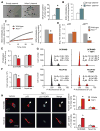
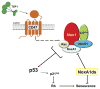
Senescent cells withdraw from the cell cycle and do not proliferate. The prevalence of senescent compared to normally functioning parenchymal cells increases with age, impairing tissue and organ homeostasis. A contentious principle governing this process has been the redox theory of aging. We linked matricellular protein thrombospondin 1 (TSP1) and its receptor CD47 to the activation of NADPH oxidase 1 (Nox1), but not of the other closely related Nox isoforms, and associated oxidative stress, and to senescence in human cells and aged tissue. In human endothelial cells, TSP1 promoted senescence and attenuated cell cycle progression and proliferation. At the molecular level, TSP1 increased Nox1-dependent generation of reactive oxygen species (ROS), leading to the increased abundance of the transcription factor p53. p53 mediated a DNA damage response that led to senescence through Rb and p21cip, both of which inhibit cell cycle progression. Nox1 inhibition blocked the ability of TSP1 to increase p53 nuclear localization and p21cip abundance and its ability to promote senescence. Mice lacking TSP1 showed decreases in ROS production, p21cip expression, p53 activity, and aging-induced senescence. Conversely, lung tissue from aging humans displayed increases in the abundance of vascular TSP1, Nox1, p53, and p21cip Finally, genetic ablation or pharmacological blockade of Nox1 in human endothelial cells attenuated TSP1-mediated ROS generation, restored cell cycle progression, and protected against senescence. Together, our results provide insights into the functional interplay between TSP1 and Nox1 in the regulation of endothelial senescence and suggest potential targets for controlling the aging process at the molecular level.
Copyright © 2017 The Authors, some rights reserved; exclusive licensee American Association for the Advancement of Science. No claim to original U.S. Government Works.
Conflict of interest statement
Figures



References
-
- Gellie A, Mills A, Levinson M, Stephenson G, Flynn E. Death: A foe to be conquered? Questioning the paradigm. Age Ageing. 2015;44:7–10. - PubMed
-
- Benetos A, Rossignol P, Cherubini A, Joly L, Grodzicki T, Rajkumar C, Strandberg TE, Petrovic M. Polypharmacy in the aging patient: Management of hypertension in octogenarians. JAMA. 2015;314:170–180. - PubMed
MeSH terms
Substances
Grants and funding
- R42 AA024003/AA/NIAAA NIH HHS/United States
- T32 GM008424/GM/NIGMS NIH HHS/United States
- K23 HL112848/HL/NHLBI NIH HHS/United States
- 15SDG24910003/AHA/American Heart Association-American Stroke Association/United States
- R01 HL108954/HL/NHLBI NIH HHS/United States
- R21 EB017184/EB/NIBIB NIH HHS/United States
- R01 HL098032/HL/NHLBI NIH HHS/United States
- R01 HL079207/HL/NHLBI NIH HHS/United States
- T32 HL007563/HL/NHLBI NIH HHS/United States
- T32 HL110849/HL/NHLBI NIH HHS/United States
- P01 HL103455/HL/NHLBI NIH HHS/United States
- R01 HL112914/HL/NHLBI NIH HHS/United States
- R01 HL123766/HL/NHLBI NIH HHS/United States
- R01 HL125886/HL/NHLBI NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

