Glucose feeds the TCA cycle via circulating lactate
- PMID: 29045397
- PMCID: PMC5898814
- DOI: 10.1038/nature24057
Glucose feeds the TCA cycle via circulating lactate
Abstract
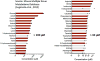
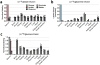
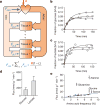
Mammalian tissues are fuelled by circulating nutrients, including glucose, amino acids, and various intermediary metabolites. Under aerobic conditions, glucose is generally assumed to be burned fully by tissues via the tricarboxylic acid cycle (TCA cycle) to carbon dioxide. Alternatively, glucose can be catabolized anaerobically via glycolysis to lactate, which is itself also a potential nutrient for tissues and tumours. The quantitative relevance of circulating lactate or other metabolic intermediates as fuels remains unclear. Here we systematically examine the fluxes of circulating metabolites in mice, and find that lactate can be a primary source of carbon for the TCA cycle and thus of energy. Intravenous infusions of 13C-labelled nutrients reveal that, on a molar basis, the circulatory turnover flux of lactate is the highest of all metabolites and exceeds that of glucose by 1.1-fold in fed mice and 2.5-fold in fasting mice; lactate is made primarily from glucose but also from other sources. In both fed and fasted mice, 13C-lactate extensively labels TCA cycle intermediates in all tissues. Quantitative analysis reveals that during the fasted state, the contribution of glucose to tissue TCA metabolism is primarily indirect (via circulating lactate) in all tissues except the brain. In genetically engineered lung and pancreatic cancer tumours in fasted mice, the contribution of circulating lactate to TCA cycle intermediates exceeds that of glucose, with glutamine making a larger contribution than lactate in pancreatic cancer. Thus, glycolysis and the TCA cycle are uncoupled at the level of lactate, which is a primary circulating TCA substrate in most tissues and tumours.
Conflict of interest statement
Figures


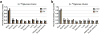






Comment in
-
Waste Not, Want Not: Lactate Oxidation Fuels the TCA Cycle.Cell Metab. 2017 Dec 5;26(6):803-804. doi: 10.1016/j.cmet.2017.11.005. Cell Metab. 2017. PMID: 29211977
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

