Loss of Protein Kinase Novel 1 (PKN1) is associated with mild systolic and diastolic contractile dysfunction, increased phospholamban Thr17 phosphorylation, and exacerbated ischaemia-reperfusion injury
- PMID: 29045568
- PMCID: PMC5815577
- DOI: 10.1093/cvr/cvx206
Loss of Protein Kinase Novel 1 (PKN1) is associated with mild systolic and diastolic contractile dysfunction, increased phospholamban Thr17 phosphorylation, and exacerbated ischaemia-reperfusion injury
Abstract
Aims: PKN1 is a stress-responsive protein kinase acting downstream of small GTP-binding proteins of the Rho/Rac family. The aim was to determine its role in endogenous cardioprotection.
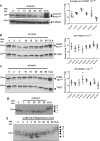
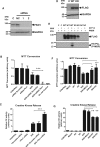
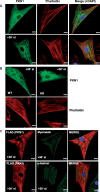
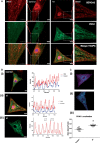
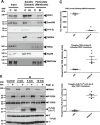
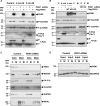
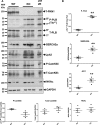
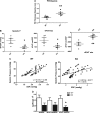
Methods and results: Hearts from PKN1 knockout (KO) or wild type (WT) littermate control mice were perfused in Langendorff mode and subjected to global ischaemia and reperfusion (I/R). Myocardial infarct size was doubled in PKN1 KO hearts compared to WT hearts. PKN1 was basally phosphorylated on the activation loop Thr778 PDK1 target site which was unchanged during I/R. However, phosphorylation of p42/p44-MAPK was decreased in KO hearts at baseline and during I/R. In cultured neonatal rat ventricular cardiomyocytes (NRVM) and NRVM transduced with kinase dead (KD) PKN1 K644R mutant subjected to simulated ischaemia/reperfusion (sI/R), PhosTag® gel analysis showed net dephosphorylation of PKN1 during sI and early R despite Thr778 phosphorylation. siRNA knockdown of PKN1 in NRVM significantly decreased cell survival and increased cell injury by sI/R which was reversed by WT- or KD-PKN1 expression. Confocal immunofluorescence analysis of PKN1 in NRVM showed increased localization to the sarcoplasmic reticulum (SR) during sI. GC-MS/MS and immunoblot analysis of PKN1 immunoprecipitates following sI/R confirmed interaction with CamKIIδ. Co-translocation of PKN1 and CamKIIδ to the SR/membrane fraction during sI correlated with phospholamban (PLB) Thr17 phosphorylation. siRNA knockdown of PKN1 in NRVM resulted in increased basal CamKIIδ activation and increased PLB Thr17 phosphorylation only during sI. In vivo PLB Thr17 phosphorylation, Sarco-Endoplasmic Reticulum Ca2+ ATPase (SERCA2) expression and Junctophilin-2 (Jph2) expression were also basally increased in PKN1 KO hearts. Furthermore, in vivo P-V loop analysis of the beat-to-beat relationship between rate of LV pressure development or relaxation and end diastolic P (EDP) showed mild but significant systolic and diastolic dysfunction with preserved ejection fraction in PKN1 KO hearts.
Conclusion: Loss of PKN1 in vivo significantly reduces endogenous cardioprotection and increases myocardial infarct size following I/R injury. Cardioprotection by PKN1 is associated with reduced CamKIIδ-dependent PLB Thr17 phosphorylation at the SR and therefore may stabilize the coupling of SR Ca2+ handling and contractile function, independent of its kinase activity.
Keywords: Protein kinase Novel 1 (PKN1) • Cardioprotection • Infarction • CamKIIδ • Phospholamban.
© The Author 2017 Published by Oxford University Press on behalf of the European Society of Cardiology.
Figures


Similar articles
-
Ablation of phospholamban rescues reperfusion arrhythmias but exacerbates myocardium infarction in hearts with Ca2+/calmodulin kinase II constitutive phosphorylation of ryanodine receptors.Cardiovasc Res. 2019 Mar 1;115(3):556-569. doi: 10.1093/cvr/cvy213. Cardiovasc Res. 2019. PMID: 30169578 Free PMC article.
-
Kcnj11 Ablation Is Associated With Increased Nitro-Oxidative Stress During Ischemia-Reperfusion Injury: Implications for Human Ischemic Cardiomyopathy.Circ Heart Fail. 2017 Feb;10(2):e003523. doi: 10.1161/CIRCHEARTFAILURE.116.003523. Circ Heart Fail. 2017. PMID: 28209764 Free PMC article.
-
Taxol, a microtubule stabilizer, improves cardiac functional recovery during postischemic reperfusion in rat in vitro.Cardiovasc Ther. 2012 Feb;30(1):12-30. doi: 10.1111/j.1755-5922.2010.00163.x. Epub 2010 Jun 14. Cardiovasc Ther. 2012. PMID: 20553295
-
Adiponectin regulates SR Ca(2+) cycling following ischemia/reperfusion via sphingosine 1-phosphate-CaMKII signaling in mice.J Mol Cell Cardiol. 2014 Sep;74:183-92. doi: 10.1016/j.yjmcc.2014.05.010. Epub 2014 May 20. J Mol Cell Cardiol. 2014. PMID: 24852843
-
The importance of the Thr17 residue of phospholamban as a phosphorylation site under physiological and pathological conditions.Braz J Med Biol Res. 2006 May;39(5):563-72. doi: 10.1590/s0100-879x2006000500001. Epub 2006 Apr 20. Braz J Med Biol Res. 2006. PMID: 16648892 Review.
Cited by
-
Intravenously transplanted mesenchymal stromal cells: a new endocrine reservoir for cardioprotection.Stem Cell Res Ther. 2022 Jun 17;13(1):253. doi: 10.1186/s13287-022-02922-z. Stem Cell Res Ther. 2022. PMID: 35715868 Free PMC article.
-
Verteporfin treatment controls morphology, phenotype, and global gene expression for cells of the human nucleus pulposus.JOR Spine. 2020 Jul 30;3(4):e1111. doi: 10.1002/jsp2.1111. eCollection 2020 Dec. JOR Spine. 2020. PMID: 33392449 Free PMC article.
-
IGF-1C domain-modified hydrogel enhanced the efficacy of stem cells in the treatment of AMI.Stem Cell Res Ther. 2020 Mar 26;11(1):136. doi: 10.1186/s13287-020-01637-3. Stem Cell Res Ther. 2020. PMID: 32216819 Free PMC article.
-
Protein Kinase N1 Level Predicts Acute Kidney Injury in Patients Undergoing Cardiac Surgery: A Prospective Cohort Study.Blood Purif. 2024;53(6):465-475. doi: 10.1159/000536225. Epub 2024 Jan 16. Blood Purif. 2024. PMID: 38228111 Free PMC article.
-
PKC and PKN in heart disease.J Mol Cell Cardiol. 2019 Mar;128:212-226. doi: 10.1016/j.yjmcc.2019.01.029. Epub 2019 Feb 8. J Mol Cell Cardiol. 2019. PMID: 30742812 Free PMC article. Review.
References
-
- Simkhovich BZ, Przyklenk K, Kloner RA.. Role of protein kinase C in ischemic ‘conditioning’: from first evidence to current perspectives. J Cardiovasc Pharmacol Ther 2013;18:525–532. - PubMed
-
- Ping P, Zhang J, Cao X, Li RC, Kong D, Tang XL, Qiu Y, Manchikalapudi S, Auchampach JA, Black RG, Bolli R.. PKC-dependent activation of p44/p42 MAPKs during myocardial ischemia-reperfusion in conscious rabbits. Am J Physiol 1999;276:H1468–H1481. - PubMed
-
- Mockridge JW, Marber MS, Heads RJ.. Activation of Akt during simulated ischemia/reperfusion in cardiac myocytes. Biochem Biophys Res Commun 2000;270:947–952. - PubMed
-
- Hausenloy DJ, Mocanu MM, Yellon DM.. Cross-talk between the survival kinases during early reperfusion: its contribution to ischemic preconditioning. Cardiovasc Res 2004;63:305–312. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

