Monocyte-Macrophages and T Cells in Atherosclerosis
- PMID: 29045897
- PMCID: PMC5747297
- DOI: 10.1016/j.immuni.2017.09.008
Monocyte-Macrophages and T Cells in Atherosclerosis
Abstract
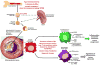
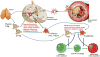
Atherosclerosis is an arterial disease process characterized by the focal subendothelial accumulation of apolipoprotein-B-containing lipoproteins, immune and vascular wall cells, and extracellular matrix. The lipoproteins acquire features of damage-associated molecular patterns and trigger first an innate immune response, dominated by monocyte-macrophages, and then an adaptive immune response. These inflammatory responses often become chronic and non-resolving and can lead to arterial damage and thrombosis-induced organ infarction. The innate immune response is regulated at various stages, from hematopoiesis to monocyte changes and macrophage activation. The adaptive immune response is regulated primarily by mechanisms that affect the balance between regulatory and effector T cells. Mechanisms related to cellular cholesterol, phenotypic plasticity, metabolism, and aging play key roles in affecting these responses. Herein, we review select topics that shed light on these processes and suggest new treatment strategies.
Keywords: Innate and adaptive inflammatory responses drive the progression of atherosclerosis; T cell; adaptive immunity; effector T cell; inflammation; innate immunity; macrophage; monocyte; regulatory T cell; which is the root cause of most cardiovascular disease. Tabas and Lichtman review the roles and regulation of monocyte-macrophages and T cells during the various stages of atherosclerosis and discuss how this knowledge suggests new therapeutic approaches.atherosclerosis.
Copyright © 2017 Elsevier Inc. All rights reserved.
Figures

References
-
- Ait-Oufella H, Pouresmail V, Simon T, Blanc-Brude O, Kinugawa K, Merval R, Offenstadt G, Leseche G, Cohen PL, Tedgui A, et al. Defective mer receptor tyrosine kinase signaling in bone marrow cells promotes apoptotic cell accumulation and accelerates atherosclerosis. Arterioscler Thromb Vasc Biol. 2008;28:1429–1431. - PubMed
-
- American Heart Association & RTI International. Cardiovascular disease: a costly burden for America - projections through 2035 2015
-
- Amit I, Winter DR, Jung S. The role of the local environment and epigenetics in shaping macrophage identity and their effect on tissue homeostasis. Nat Immunol. 2016;17:18–25. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

