Soluble CD40 Ligand and Oxidative Response Are Reciprocally Stimulated during Shiga Toxin-Associated Hemolytic Uremic Syndrome
- PMID: 29068360
- PMCID: PMC5705951
- DOI: 10.3390/toxins9110331
Soluble CD40 Ligand and Oxidative Response Are Reciprocally Stimulated during Shiga Toxin-Associated Hemolytic Uremic Syndrome
Abstract
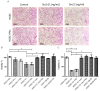
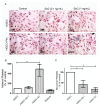
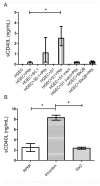
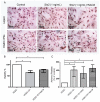
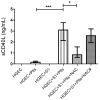
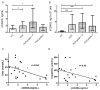
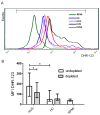
Shiga toxin (Stx), produced by Escherichia coli, is the main pathogenic factor of diarrhea-associated hemolytic uremic syndrome (HUS), which is characterized by the obstruction of renal microvasculature by platelet-fibrin thrombi. It is well known that the oxidative imbalance generated by Stx induces platelet activation, contributing to thrombus formation. Moreover, activated platelets release soluble CD40 ligand (sCD40L), which in turn contributes to oxidative imbalance, triggering the release of reactive oxidative species (ROS) on various cellular types. The aim of this work was to determine if the interaction between the oxidative response and platelet-derived sCD40L, as consequence of Stx-induced endothelium damage, participates in the pathogenic mechanism during HUS. Activated human glomerular endothelial cells (HGEC) by Stx2 induced platelets to adhere to them. Although platelet adhesion did not contribute to endothelial damage, high levels of sCD40L were released to the medium. The release of sCD40L by activated platelets was inhibited by antioxidant treatment. Furthermore, we found increased levels of sCD40L in plasma from HUS patients, which were also able to trigger the respiratory burst in monocytes in a sCD40L-dependent manner. Thus, we concluded that platelet-derived sCD40L and the oxidative response are reciprocally stimulated during Stx2-associated HUS. This process may contribute to the evolution of glomerular occlusion and the microangiopathic lesions.
Keywords: CD40L; Shiga toxin 2; blood platelets; hemolytic uremic syndrome; oxidative stress.
Conflict of interest statement
The authors declare that they have no conflict of interest.
Figures
Similar articles
-
Pathogenic role of inflammatory response during Shiga toxin-associated hemolytic uremic syndrome (HUS).Pediatr Nephrol. 2018 Nov;33(11):2057-2071. doi: 10.1007/s00467-017-3876-0. Epub 2018 Jan 25. Pediatr Nephrol. 2018. PMID: 29372302 Review.
-
Platelet activation by Shiga toxin and circulatory factors as a pathogenetic mechanism in the hemolytic uremic syndrome.Blood. 2001 May 15;97(10):3100-8. doi: 10.1182/blood.v97.10.3100. Blood. 2001. PMID: 11342436
-
Shiga toxin triggers endothelial and podocyte injury: the role of complement activation.Pediatr Nephrol. 2019 Mar;34(3):379-388. doi: 10.1007/s00467-017-3850-x. Epub 2017 Dec 6. Pediatr Nephrol. 2019. PMID: 29214442 Review.
-
Hemolytic uremic syndrome-associated Shiga toxins promote endothelial-cell secretion and impair ADAMTS13 cleavage of unusually large von Willebrand factor multimers.Blood. 2005 Dec 15;106(13):4199-209. doi: 10.1182/blood-2005-05-2111. Epub 2005 Aug 30. Blood. 2005. PMID: 16131569 Free PMC article.
-
Shiga toxin-induced complement-mediated hemolysis and release of complement-coated red blood cell-derived microvesicles in hemolytic uremic syndrome.J Immunol. 2015 Mar 1;194(5):2309-18. doi: 10.4049/jimmunol.1402470. Epub 2015 Jan 30. J Immunol. 2015. PMID: 25637016
Cited by
-
CD40/CD40L Signaling as a Promising Therapeutic Target for the Treatment of Renal Disease.J Clin Med. 2020 Nov 13;9(11):3653. doi: 10.3390/jcm9113653. J Clin Med. 2020. PMID: 33202988 Free PMC article. Review.
-
Lentiviral gene therapy corrects platelet phenotype and function in patients with Wiskott-Aldrich syndrome.J Allergy Clin Immunol. 2019 Sep;144(3):825-838. doi: 10.1016/j.jaci.2019.03.012. Epub 2019 Mar 27. J Allergy Clin Immunol. 2019. PMID: 30926529 Free PMC article.
-
Recent Advances in Shiga Toxin-Producing Escherichia coli Research in Latin America.Microorganisms. 2018 Sep 28;6(4):100. doi: 10.3390/microorganisms6040100. Microorganisms. 2018. PMID: 30274180 Free PMC article. Review.
References
-
- O’Brien A.D., Tesh V.L., Donohue-Rolfe A., Jackson M.P., Olsnes S., Sandvig K., Lindberg A.A., Keusch G.T. Shiga toxin: Biochemistry, genetics, mode of action, and role in pathogenesis. Curr. Top. Microbiol. Immunol. 1992;180:65–94. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

