Efficacy and safety of immunosuppressive medications for steroid-resistant nephrotic syndrome in children: a systematic review and network meta-analysis
- PMID: 29069848
- PMCID: PMC5641191
- DOI: 10.18632/oncotarget.20377
Efficacy and safety of immunosuppressive medications for steroid-resistant nephrotic syndrome in children: a systematic review and network meta-analysis
Abstract
Background: Conventional meta-analyses and randomized controlled trials have shown inconsistent results regarding the efficacy of immunosuppressants for pediatric steroid-resistant nephrotic syndrome (SRNS).
Objective: To conduct a network meta-analysis aimed at evaluating the efficacy and safety of available immunosuppressive agents in pediatric patients with SRNS.
Study methods: MEDLINE, the Cochrane Central Register of Controlled Trials, and EMBASE were searched on January 2017. Data from randomized controlled trials (RCTs) were included. The main outcomes analyzed were efficacy [number/portion with complete remission (CR), number/portion with partial remission (PR), and total number/portion in remission (TR)] and safety [adverse secondary event (ASE) rates].
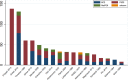
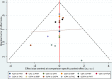
Results: A meta-analysis of 18 RCTs showed that tacrolimus was more efficacious for achieving CR than intravenous (i.v.) cyclophosphamide, mycophenolate mofetil (MMF), oral cyclophosphamide, leflunomide, chlorambucil, azathioprine, and plaebo/nontreatment (P/NT), and more efficacious than i.v. cyclophosphamide, oral cyclophosphamide, and P/NT in terms of TR outcomes. Cyclosporin was associated with a greater CR rate than i.v. cyclophosphamide, MMF, oral cyclophosphamide, chlorambucil, azathioprine, or P/NT, and associated with a greater TR rate than i.v. cyclophosphamide, oral cyclophosphamide, or P/NT. MMF was found to be more efficacious than i.v. cyclophosphamide and oral cyclophosphamide in terms of TR.
Conclusions: Tacrolimus and cyclosporine may be preferred initial treatments for children with SRNS. MMF may be another option for this patient population. Further studies of the efficacy and safety of these three drugs in children with SRNS should be pursued.
Keywords: SRNS; immunosuppressant; multiple-treatments meta-analysis; pediatrics.
Conflict of interest statement
CONFLICTS OF INTEREST The authors declare no conflicts of interest concerning this article.
Figures





References
-
- McKinney PA, Feltbower RG, Brocklebank JT, Fitzpatrick MM. Time trends and ethnic patterns of childhood nephrotic syndrome in Yorkshire, UK. Pediatr Nephrol. 2001;16:1040–1044. - PubMed
-
- Beck L, Bomback AS, Choi MJ, Holzman LB, Langford C, Mariani LH, Somers MJ, Trachtman H, Waldman M. KDOQI US commentary on the 2012 KDIGO clinical practice guideline for glomerulonephritis. Am J Kidney Dis. 2013;62:403–441. - PubMed
-
- Prospective, controlled trial of cyclophosphamide therapy in children with nephrotic syndrome Report of the International study of Kidney Disease in Children. Lancet. 1974;2:423–427. - PubMed
-
- Tune BM, Lieberman E, Mendoza SA. Steroid-resistant nephrotic focal segmental glomerulosclerosis: a treatable disease. Pediatr Nephrol. 1996;10:772–778. - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

