Error-Prone and Error-Free Translesion DNA Synthesis over Site-Specifically Created DNA Adducts of Aryl Hydrocarbons (3-Nitrobenzanthrone and 4-Aminobiphenyl)
- PMID: 29071010
- PMCID: PMC5654197
- DOI: 10.5487/TR.2017.33.4.265
Error-Prone and Error-Free Translesion DNA Synthesis over Site-Specifically Created DNA Adducts of Aryl Hydrocarbons (3-Nitrobenzanthrone and 4-Aminobiphenyl)
Abstract
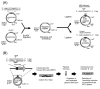
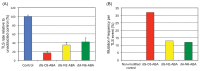
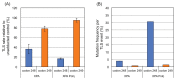
Aryl hydrocarbons such as 3-nitrobenzanthrone (NBA), 4-aminobiphenyl (ABP), acetylaminofluorene (AAF), benzo(a)pyrene (BaP), and 1-nitropyrene (NP) form bulky DNA adducts when absorbed by mammalian cells. These chemicals are metabolically activated to reactive forms in mammalian cells and preferentially get attached covalently to the N2 or C8 positions of guanine or the N6 position of adenine. The proportion of N2 and C8 guanine adducts in DNA differs among chemicals. Although these adducts block DNA replication, cells have a mechanism allowing to continue replication by bypassing these adducts: translesion DNA synthesis (TLS). TLS is performed by translesion DNA polymerases-Pol η, κ, ι, and ζ and Rev1-in an error-free or error-prone manner. Regarding the NBA adducts, namely, 2-(2'-deoxyguanosin-N2-yl)-3-aminobenzanthrone (dG-N2-ABA) and N-(2'-deoxyguanosin-8-yl)-3-aminobenzanthrone (dG-C8-ABA), dG-N2-ABA is produced more often than dG-C8-ABA, whereas dG-C8-ABA blocks DNA replication more strongly than dG-N2-ABA. dG-N2-ABA allows for a less error-prone bypass than dG-C8-ABA does. Pol η and κ are stronger contributors to TLS over dG-C8-ABA, and Pol κ bypasses dG-C8-ABA in an error-prone manner. TLS efficiency and error-proneness are affected by the sequences surrounding the adduct, as demonstrated in our previous study on an ABP adduct, N-(2'-deoxyguanosine-8-yl)-4-aminobiphenyl (dG-C8-ABP). Elucidation of the general mechanisms determining efficiency, error-proneness, and the polymerases involved in TLS over various adducts is the next step in the research on TLS. These TLS studies will clarify the mechanisms underlying aryl hydrocarbon mutagenesis and carcinogenesis in more detail.
Keywords: 3-Nitrobenzanthrone; Aryl hydrocarbon; DNA adduct; Mutation; Translesion DNA synthesis.
Conflict of interest statement
CONFLICT OF INTEREST The authors declare that they have no competing interests.
Figures

Similar articles
-
Mutational analysis of the C8-guanine adduct of the environmental carcinogen 3-nitrobenzanthrone in human cells: critical roles of DNA polymerases η and κ and Rev1 in error-prone translesion synthesis.Biochemistry. 2014 Aug 19;53(32):5323-31. doi: 10.1021/bi5007805. Epub 2014 Aug 6. Biochemistry. 2014. PMID: 25080294 Free PMC article.
-
Translesion DNA synthesis across various DNA adducts produced by 3-nitrobenzanthrone in Escherichia coli.Mutat Res. 2013 Jun 14;754(1-2):32-8. doi: 10.1016/j.mrgentox.2013.04.001. Epub 2013 Apr 9. Mutat Res. 2013. PMID: 23583687
-
Adduct formation and repair, and translesion DNA synthesis across the adducts in human cells exposed to 3-nitrobenzanthrone.Mutat Res. 2013 May 15;753(2):93-100. doi: 10.1016/j.mrgentox.2013.03.005. Epub 2013 Apr 6. Mutat Res. 2013. PMID: 23567330
-
Translesion synthesis in Escherichia coli: lessons from the NarI mutation hot spot.DNA Repair (Amst). 2007 Jul 1;6(7):1032-41. doi: 10.1016/j.dnarep.2007.02.021. Epub 2007 Apr 2. DNA Repair (Amst). 2007. PMID: 17403618 Review.
-
Filling gaps in translesion DNA synthesis in human cells.Mutat Res Genet Toxicol Environ Mutagen. 2018 Dec;836(Pt B):127-142. doi: 10.1016/j.mrgentox.2018.02.004. Epub 2018 Feb 23. Mutat Res Genet Toxicol Environ Mutagen. 2018. PMID: 30442338 Review.
Cited by
-
Nitroreduction: A Critical Metabolic Pathway for Drugs, Environmental Pollutants, and Explosives.Chem Res Toxicol. 2022 Oct 17;35(10):1747-1765. doi: 10.1021/acs.chemrestox.2c00175. Epub 2022 Aug 31. Chem Res Toxicol. 2022. PMID: 36044734 Free PMC article. Review.
-
EGFP Reporters for Direct and Sensitive Detection of Mutagenic Bypass of DNA Lesions.Biomolecules. 2020 Jun 13;10(6):902. doi: 10.3390/biom10060902. Biomolecules. 2020. PMID: 32545792 Free PMC article.
-
Origin of mutations in genes associated with human glioblastoma multiform cancer: random polymerase errors versus deamination.Heliyon. 2019 Mar 7;5(3):e01265. doi: 10.1016/j.heliyon.2019.e01265. eCollection 2019 Mar. Heliyon. 2019. PMID: 30899826 Free PMC article.
-
Mammalian DNA Polymerase Kappa Activity and Specificity.Molecules. 2019 Aug 1;24(15):2805. doi: 10.3390/molecules24152805. Molecules. 2019. PMID: 31374881 Free PMC article. Review.
-
Probing the Effect of Bulky Lesion-Induced Replication Fork Conformational Heterogeneity Using 4-Aminobiphenyl-Modified DNA.Molecules. 2019 Apr 20;24(8):1566. doi: 10.3390/molecules24081566. Molecules. 2019. PMID: 31009995 Free PMC article.
References
-
- Choi H, Harrison R, Komulainen H, Saborit JMD. WHO Guidelines for Indoor Air Quality: Selected Pollutants. World Health Organization; Geneva: 2010. Polycyclic aromatic hydrocarbons; pp. 299–346.
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

