Kinetic characterization of the inhibition of protein tyrosine phosphatase-1B by Vanadyl (VO2+) chelates
- PMID: 29071441
- PMCID: PMC5671894
- DOI: 10.1007/s00775-017-1500-1
Kinetic characterization of the inhibition of protein tyrosine phosphatase-1B by Vanadyl (VO2+) chelates
Abstract
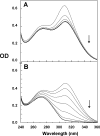
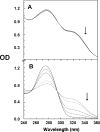
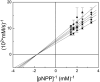
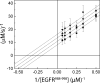
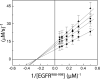
Protein tyrosine phosphatases (PTPases) are a prominent focus of drug design studies because of their roles in homeostasis and disorders of metabolism. These studies have met with little success because (1) virtually all inhibitors hitherto exhibit only competitive behavior and (2) a consensus sequence H/V-C-X5-R-S/T characterizes the active sites of PTPases, leading to low specificity of active site directed inhibitors. With protein tyrosine phosphatase-1B (PTP1B) identifed as the target enzyme of the vanadyl (VO2+) chelate bis(acetylacetonato)oxidovanadium(IV) [VO(acac)2] in 3T3-L1 adipocytes [Ou et al. J Biol Inorg Chem 10: 874-886, 2005], we compared the inhibition of PTP1B by VO(acac)2 with other VO2+-chelates, namely, bis(2-ethyl-maltolato)oxidovanadium(IV) [VO(Et-malto)2] and bis(3-hydroxy-2-methyl-4(1H)pyridinonato)oxidovanadium(IV) [VO(mpp)2] under steady-state conditions, using the soluble portion of the recombinant human enzyme (residues 1-321). Our results differed from those of previous investigations because we compared inhibition in the presence of the nonspecific substrate p-nitrophenylphosphate and the phosphotyrosine-containing undecapeptide DADEpYLIPQQG mimicking residues 988-998 of the epidermal growth factor receptor, a relevant, natural substrate. While VO(Et-malto)2 acts only as a noncompetitive inhibitor in the presence of either subtrate, VO(acac)2 exhibits classical uncompetitive inhibition in the presence of DADEpYLIPQQG but only apparent competitive inhibition with p-nitrophenylphosphate as substrate. Because uncompetitive inhibitors are more potent pharmacologically than competitive inhibitors, structural characterization of the site of uncompetitive binding of VO(acac)2 may provide a new direction for design of inhibitors for therapeutic purposes. Our results suggest also that the true behavior of other inhibitors may have been masked when assayed with only p-nitrophenylphosphate as substrate.
Keywords: Bis(acetylacetonato)oxidovanadium(IV); Protein tyrosine phosphatase-1B; Steady-state kinetics; Uncompetitive inhibition; VO(acac)2; Vanadyl (VO2+) chelates.
Figures

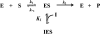
References
-
- Tonks NK, Diltz CD, Fischer EH. J Biol Chem. 1988;263:6731–67372. - PubMed
-
- Tonks NK, Diltz CD, Fischer EH. J Biol Chem. 1988;263:6722–6730. - PubMed
-
- Sarmiento M, Zhao Y, Gordon SJ, Zhang ZY. J Biol Chem. 1998;273:26368–2637. - PubMed
-
- Zhang S, Zhang ZY. Drug Discovery Today. 2007;12:373–381. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

