Synthesis and biological evaluation of histamine Schiff bases as carbonic anhydrase I, II, IV, VII, and IX activators
- PMID: 29072105
- PMCID: PMC6010137
- DOI: 10.1080/14756366.2017.1386660
Synthesis and biological evaluation of histamine Schiff bases as carbonic anhydrase I, II, IV, VII, and IX activators
Abstract
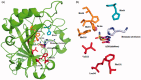
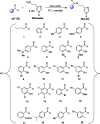
A series of 20 histamine Schiff base was synthesised by reaction of histamine, a well known carbonic anhydrase (CA, E.C 4.2.2.1.) activator pharmacophore, with substituted aldehydes. The obtained histamine Schiff bases were assayed as activators of five selected human (h) CA isozymes, the cytosolic hCA I, hCA II, and hCA VII, the membrane-anchored hCA IV and transmembrane hCA IX. Some of these compounds showed efficient activity (in the nanomolar range) against the cytosolic isoform hCA VII, which is a key CA enzyme involved in brain metabolism. Moderate activity was observed against hCA I and hCA IV (in the nanomolar to low micromolar range). The structure-activity relationship for activation of these isoforms with the new histamine Schiff bases is discussed in detail based on the nature of the aliphatic, aromatic, or heterocyclic moiety present in the aldehyde fragment of the molecule, which may participate in diverse interactions with amino acid residues at the entrance of the active site, where activators bind, and which is the most variable part among the different CA isoforms.
Keywords: Alzheimer’s disease; Carbonic anhydrase activators; Schiff bases; histamine; isozymes.
Figures
Similar articles
-
Synthesis and biological evaluation of novel aromatic and heterocyclic bis-sulfonamide Schiff bases as carbonic anhydrase I, II, VII and IX inhibitors.Bioorg Med Chem. 2017 Jun 15;25(12):3093-3097. doi: 10.1016/j.bmc.2017.03.063. Epub 2017 Mar 31. Bioorg Med Chem. 2017. PMID: 28400084
-
Inhibition of carbonic anhydrase isoforms I, II, IX and XII with novel Schiff bases: identification of selective inhibitors for the tumor-associated isoforms over the cytosolic ones.Bioorg Med Chem. 2014 Nov 1;22(21):5883-90. doi: 10.1016/j.bmc.2014.09.021. Epub 2014 Sep 21. Bioorg Med Chem. 2014. PMID: 25267005
-
Pyridinium derivatives of histamine are potent activators of cytosolic carbonic anhydrase isoforms I, II and VII.Org Biomol Chem. 2011 Apr 21;9(8):2790-800. doi: 10.1039/c0ob00703j. Epub 2011 Mar 2. Org Biomol Chem. 2011. PMID: 21369613
-
Amine- and Amino Acid-Based Compounds as Carbonic Anhydrase Activators.Molecules. 2021 Dec 2;26(23):7331. doi: 10.3390/molecules26237331. Molecules. 2021. PMID: 34885917 Free PMC article. Review.
-
The Effect of Nanoparticles on the Structure and Enzymatic Activity of Human Carbonic Anhydrase I and II.Molecules. 2020 Sep 25;25(19):4405. doi: 10.3390/molecules25194405. Molecules. 2020. PMID: 32992797 Free PMC article. Review.
Cited by
-
The first activation study of the β-carbonic anhydrases from the pathogenic bacteria Brucella suis and Francisella tularensis with amines and amino acids.J Enzyme Inhib Med Chem. 2019 Dec;34(1):1178-1185. doi: 10.1080/14756366.2019.1630617. J Enzyme Inhib Med Chem. 2019. PMID: 31282230 Free PMC article.
-
Synthesis and Molecular Docking of New N-Acyl Hydrazones- Benzimidazole as hCA I and II Inhibitors.Med Chem. 2023;19(5):485-494. doi: 10.2174/1573406419666221222143530. Med Chem. 2023. PMID: 36567289 Free PMC article.
-
Carbonic Anhydrase Activators for Neurodegeneration: An Overview.Molecules. 2022 Apr 14;27(8):2544. doi: 10.3390/molecules27082544. Molecules. 2022. PMID: 35458743 Free PMC article. Review.
-
A structure-based approach towards the identification of novel antichagasic compounds: Trypanosoma cruzi carbonic anhydrase inhibitors.J Enzyme Inhib Med Chem. 2020 Dec;35(1):21-30. doi: 10.1080/14756366.2019.1677638. J Enzyme Inhib Med Chem. 2020. PMID: 31619095 Free PMC article.
-
New Histamine-Related Five-Membered N-Heterocycle Derivatives as Carbonic Anhydrase I Activators.Molecules. 2022 Jan 15;27(2):545. doi: 10.3390/molecules27020545. Molecules. 2022. PMID: 35056859 Free PMC article.
References
-
- Supuran CT.Advances in structure-based drug discovery of carbonic anhydrase inhibitors. Expert Opin Drug Discov 2017;12:61–88. - PubMed
-
- Supuran CT.How many carbonic anhydrase inhibition mechanisms exist? J Enzym Inhib Med Chem 2016;31:345–60. - PubMed
-
- Supuran CT.Structure and function of carbonic anhydrases. Biochem J 2016;473:2023–32. - PubMed
-
- Supuran CT.Carbonic anhydrases: novel therapeutic applications for inhibitors and activators. Nat Rev Drug Discov 2008;7:168–81. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
