Concise Review: Cancer Cells, Cancer Stem Cells, and Mesenchymal Stem Cells: Influence in Cancer Development
- PMID: 29072369
- PMCID: PMC5702541
- DOI: 10.1002/sctm.17-0138
Concise Review: Cancer Cells, Cancer Stem Cells, and Mesenchymal Stem Cells: Influence in Cancer Development
Abstract
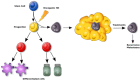
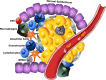
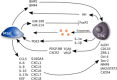
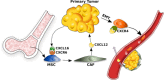
Tumors are composed of different types of cancer cells that contribute to tumor heterogeneity. Among these populations of cells, cancer stem cells (CSCs) play an important role in cancer initiation and progression. Like their stem cells counterpart, CSCs are also characterized by self-renewal and the capacity to differentiate. A particular population of CSCs is constituted by mesenchymal stem cells (MSCs) that differentiate into cells of mesodermal characteristics. Several studies have reported the potential pro-or anti-tumorigenic influence of MSCs on tumor initiation and progression. In fact, MSCs are recruited to the site of wound healing to repair damaged tissues, an event that is also associated with tumorigenesis. In other cases, resident or migrating MSCs can favor tumor angiogenesis and increase tumor aggressiveness. This interplay between MSCs and cancer cells is fundamental for cancerogenesis, progression, and metastasis. Therefore, an interesting topic is the relationship between cancer cells, CSCs, and MSCs, since contrasting reports about their respective influences have been reported. In this review, we discuss recent findings related to conflicting results on the influence of normal and CSCs in cancer development. The understanding of the role of MSCs in cancer is also important in cancer management. Stem Cells Translational Medicine 2017;6:2115-2125.
Keywords: Cancer progression; Drug resistance; Epithelial to mesenchymal transition; Mesenchymal stem cells; Microenvironment.
© 2017 The Authors Stem Cells Translational Medicine published by Wiley Periodicals, Inc. on behalf of AlphaMed Press.
Figures
References
-
- Reya T, Morrison SJ, Clarke MF et al. Stem cells, cancer, and cancer stem cells. Nature 2001;414:105–111. - PubMed
-
- Chamberlain G, Fox J, Ashton B et al. Concise review: Mesenchymal stem cells: Their phenotype, differentiation capacity, immunological features, and potential for homing. Stem Cells 2007;25:2739–2749. - PubMed
-
- Roufosse CA, Direkze NC, Otto WR et al. Circulating mesenchymal stem cells. Int J Biochem Cell Biol 2004;36:585–597. - PubMed
-
- Lee RH, Kim B, Choi I et al. Characterization and expression analysis of mesenchymal stem cells from human bone marrow and adipose tissue. Cell Physiol Biochem 2004;14:311–324. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

